Translate this page into:
Large retroperitoneal soft tissue tumor: A cytopathological diagnosis
-
Received: ,
Accepted: ,
This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
A 65-year-old male presented to the surgery outpatient department with complaints of abdominal pain, gradually increasing abdominal size and bilateral pedal edema for the past 8 months. On perabdominal examination, a large vague abdominal mass was found to be measuring approximately 30 cm × 25 cm with the upper pole extending up to the surface of liver and lower pole extending to the upper level of pelvic brim.
Further, contrast-enhanced computed tomography (CECT) abdomen revealed a large retroperitoneal mass lesion of size 31 cm × 22 cm × 20 cm with areas of fat density within and showed heterogeneous postcontrast enhancement causing displacement of right kidney, pancreas, and bowel loops, most likely suggestive of liposarcoma [Figure 1a].
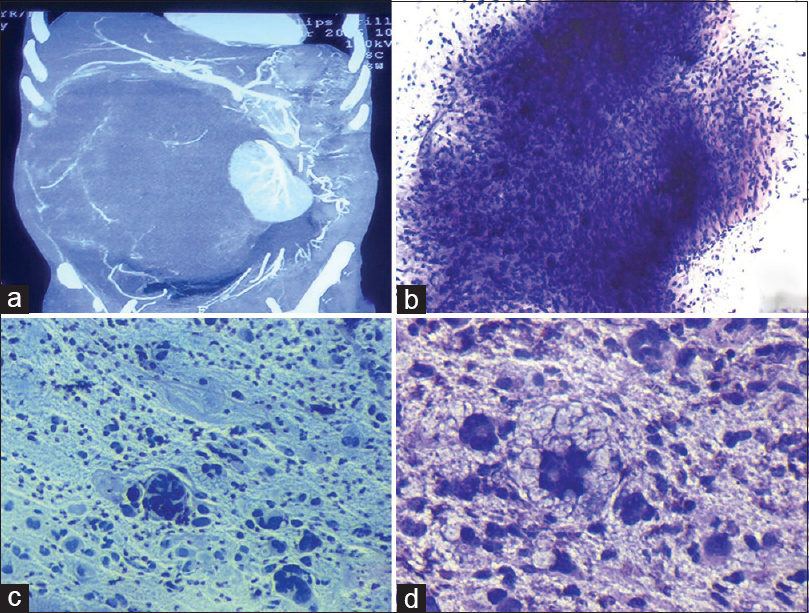
- (a) Contrast-enhanced computed tomography abdomen showing a large retroperitoneal mass lesion of size 31 cm × 22 cm × 20 cm with areas of fat density within and revealing displacement of right kidney, pancreas, and bowel loops. (b) Highly cellular smear composed of fragments of oval to spindle-shaped cells embedded in myxoid matrix (Giemsa ×100). (c) Smear showing few binucleated, multinucleated bizzare cells along with atypical mitosis and lipoblast. [Pap ×200]. (d) Atypical lipoblasts having multilobated and indented nuclei with multiple vacuoles in the cytoplasm in a myxoid background (Giemsa ×400)
Fine-needle aspiration cytology.(FNAC) of the abdominal mass was done from multiple sites which yielded thick cellular aspirate. Multiple smears prepared were air dried and fixed in 95% ethyl alcohol and subsequently stained with Giemsa and Papanicolaou stain, respectively.
QUIZ QUESTION
Q1: What is your interpretation?
-
Chordoma
-
Myxofibrosarcoma
-
Well-differentiated liposarcoma
-
Pleomorphic liposarcoma.
ANSWER
Ans 1: Option d – Pleomorphic liposarcoma (PLS).
Explanation
Smears examined showed high cellularity comprising highly pleomorphic malignant cells arranged in large sheets embedded in myxoid matrix, loosely cohesive clusters, and groups as well as many singly dispersed [Figure 1b]. The cells were round to oval and spindle-shaped with high nuclear-cytoplasmic ratio, marked anisonucleosis, irregular nuclear membrane, coarse granular chromatin, prominent nucleoli, and scant to moderate amount of pale foamy cytoplasm [Figure 1c]. Few binucleated, multinucleated bizarre tumor cells and mitosis were also noted [Figure 1c]. Many scattered atypical/pleomorphic lipoblasts were also seen having multilobated, hyperchromatic, and indented nuclei with multiple vacuoles in the cytoplasm [Figure 1d]. Few inflammatory cells and myxoid matrix were also seen in the background. FNAC diagnosis given was of pleomorphic sarcoma showing atypical/pleomorphic lipoblasts, suggestive of PLS. Option a was ruled out as in chordoma, cells are highly pleomorphic with vacuolated, eosinophilic cytoplasm and myxoid matrix. It is also important to remember that chordoma is usually noted in sacrum, clivus, and vertebral region. In myxofibrosarcoma (option b), variable cellularity is seen and tumor cells are round to spindled with variable amount of cytoplasm in a myxoid granular to filamentous background. In myxofibrosarcoma, important finding is the presence of pseudolipoblasts and is commonly found in limb and girdle. Option d), Well-differentiated liposarcoma shows lipoma-like features, with the presence of scattered pleomorphic/atypical lipoblasts and are commonly found in deep soft tissues of lower extremities.
Exploratory laparotomy was done and the tumor was excised and was sent for histopathological examination. Grossly, a large encapsulated soft tissue mass was received, measuring 30 cm × 24 cm × 18 cm [Figure 2a]. The external surface was irregular, bosselated, and covered by fascia. Cut section of tumor showed variegated appearance with gray-white firm areas forming nodules and divided by fibrous septae along with mucoid areas, yellow and hemorrhagic areas. Histopathological examination was done [Figure 2b and c].
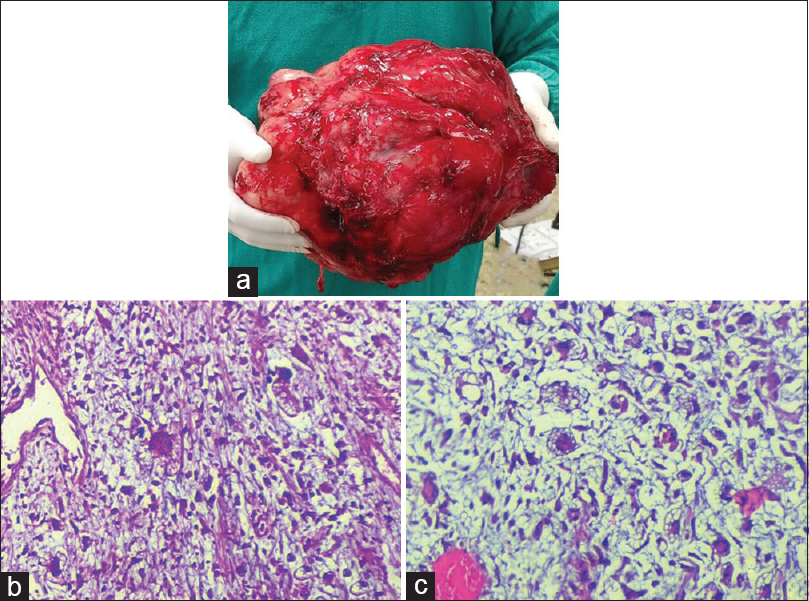
- (a) Gross image of the large encapsulated soft tissue mass, measuring 30 cm × 24 cm × 18 cm and showing irregular and bosselated surface covered by fascia. (b and c) Sections showing cells with moderate to marked pleomorphism including multinucleated cells, atypical lipoblasts in a myxoid background (H and E, ×200)
Q2: What is your interpretation after overall CECT, cytological, and histopathological examination:
-
Atypical lipoma
-
Well-differentiated liposarcoma
-
Pleomorphic Liposarcoma (PLS)
-
Myxoid liposarcoma.
Ans 2: option c – Pleopmorphic liposarcoma (PLS).
Explanation
Sections showed malignant spindle cell tumor comprised spindle cells with intervening thin-walled capillaries in a myxoid background. Also seen were cells with moderate to marked pleomorphism including many multinucleated cells with eosinophilic cytoplasm, brisk mitosis, and areas of necrosis [Figure 2b]. However, atypical/pleomorphic lipoblasts could be demonstrated only after multiple sectioning and grossing [Figure 2c]. Immunohistochemistry for S100 was negative. Histopathological diagnosis of PLS was made. The patient was given a cycle of radiotherapy and chemotherapy postoperatively and was doing well till last follow-up. However, the patient was lost to follow-up thereafter. Option (a) Atypical lipoma shows admixture of mature adipocytes and bland spindle cells along with the presence of bizarre, multinucleated cells with hyperchromatic nuclei (floret cells). Thick bright eosinophilic collagen bundles are also noted. These findings were not seen in our case. Atypical/pleomorphic lipoma (PL) is usually seen in subcutaneous tissue of posterior neck and shoulder. Option (b) well-differentiated liposarcoma was ruled out based on the findings as described earlier. This is not myxoid liposarcoma, option (d) as we did not find uniform round to oval primitive mesenchymal cells or presence of curved branching capillary vessels and myxoid stroma. However, in few cases of myxoid liposarcoma, evidence of some lipoblastic differentiation can be noted.
DISCUSSION
Liposarcoma is one of the most common soft tissue tumors with peak incidence noted in 5th-6th decade of life.[1] Usually, tumor size ranges from 5 to 10 cm; however, larger tumors have also been described as was noted in our case.[2] On CECT, the appearance of liposarcomas varies from a predominantly fat-containing lesion to a solid mass.[3] The heterogeneity of the lesion is according to the extent of necrosis, calcification, vascularity, cystic change, and soft tissue component of liposarcomas.[4]
Based on the clinicopathologic and cytogenetic studies, liposarcomas are divided into well-differentiated, dedifferentiated liposarcoma, myxoid/round cell liposarcoma, and PLS. PLS is the rarest subtype, accounting for only 5%–10% of all liposarcomas.[5] The most frequent location of PLS is lower or upper extremity. Other rare sites reported are lung, breast, retrotonsillar fossa, bone, parotid, mediastinum.[3] Morphological hallmark is the recognition of atypical/pleomorphic lipoblasts and although shows positive immunohistochemistry for S100, the latter does not play a significant role in recognizing these.
On cytology, PLS appears nondistinctive, high-grade pleomorphic, or spindle cell sarcoma. Cytomorphologically, the main diagnostic clue is the presence of atypical/pleomorphic multivacuolated lipoblasts. It should be kept in mind that aspirates from chordoma will show lipoblast-like cells, however have distinct clinical localization which avoids misdiagnosis in most instances.[5] The epithelioid variant of PLS may mimic renal cell carcinoma or adrenal cortical carcinoma.
Cytogenetics plays a significant role in subtyping the liposarcomas. Well-differentiated and dedifferentiated liposarcomas show amplification of Murine double minute 2 homolog (MDM 2) and CDK4 genes.[6789] These molecular alterations help in distinguishing well-differentiated liposarcoma from benign adipose tumors and dedifferentiated liposarcomas from other poorly differentiated sarcomas. Myxoid liposarcoma shows DNA damage - inducible transcript 3 (DD1T3) rearrangements.[6789] PLS comprise only 5% of all liposarcoma, do not show any specific aberration, and can be differentiated from myxofibrosarcoma only by the presence of pleomorphic lipoblasts.[6] However, molecular studies in the present case could not be done due to unavailability and financial constraints.
In a review by Fletcher et al.,[6] it was concluded that adequate histopathological gross sampling is essential to show the presence of lipoblastic differentiation and even a focal collection of pleomorphic lipoblasts can help in labeling the tumor as PLS. In the present case, since the tumor was very large in size, it was only after extensive grossing from various areas that revealed pleomorphic lipoblasts.
Surgical resection is the basic treatment modality for liposarcoma along with adequate resection of the margins. These tumors are more radiosensitive when compared to other sarcomas. The application of adjuvant chemotherapy has been tried in many research centers.[7] PLS has a propensity for recurrence and metastasis.[8] Recurrence rate of 15%–36% has been noted in numerous studies over a period of 10–30 years of follow-up.[9]
ADDITIONAL CME QUESTIONS
Q3: What are the specific molecular alterations in well-differentiated and dedifferentiated liposarcoma?
-
MDM2 and DD1T3
-
BRCA2 and MDM2
-
DD1T3 and CDK4
-
CDK4 and GLI.
Ans: 3a – MDM 2 homolog amplification is seen in well-differentiated and dedifferentiated liposarcoma and DNA DD1T3 rearrangements in myxoid liposarcoma. Unlike other subtypes of liposarcoma, PLS fails to show a specific molecular defect. CDK4 and GLI mutation are seen in well-differentiated liposarcoma and malignant fibrous histiocytoma.
Q4: Floret cells are usually seen in?
-
Neurofibroma
-
Myxofibrosarcoma
-
Atypical/Pleomorphic Lipoma (PL)
-
Meningioma.
Ans: 4c – Floret cells are bizarre, multinucleated cells with wreath-like hyperchromatic nuclei. These are seen in PL. PL is a circumscribed tumor with the presence of mature adipocytes admixed with spindle cells and thick bright eosinophilic collagen bundles. No lipoblasts or prominent vascularity is seen. PL shows immunoreactivity for CD34 and vimentin.
Floret cells can also be noted in neurofibromas and well-differentiated liposarcoma. These cells are not seen in myxofibrosarcoma and meningioma.
SUMMARY
Giant soft tissue tumors may be challenging, often require extensive grossing and sectioning. At times, morphology may be the key to diagnosis with limited role of immunohistochemistry. Accurate diagnosis of PLS at an unusual site can be confidently made on FNAC by demonstration of atypical/pleomorphic lipoblast.
COMPETING INTEREST STATEMENT BY ALL THE AUTHORS
The authors declare that they have no competing interest.
AUTHORSHIP STATEMENTS BY ALL AUTHORS
All authors of this article declare that we qualify for authorship as defined by ICMJE. Each author has participated sufficiently in work and takes public responsibility for appropriateness of content of this article.
ETHIC STATEMENT BY ALL AUTHORS
As this is a quiz case, this case does not require approval from the Institutional Review Board.
LIST OF ABBREVIATIONS (in alphabetic order)
-
CECT - Contrast enhanced computed tomography
-
IHC - Immunohistochemistry
-
Pap - Papanicolaou
-
PLS - Pleomorphic liposarcoma
-
FNAC - Fine needle aspiration cytology.
EDITORIAL/PEER-REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double-blind model (the authors are blinded for reviewers and vice versa) through automatic online system.
REFERENCES
- Pathology and genetics of tumours of soft tissue and bone. In: World Health Organisation Classification of Tumours (4th ed). Lyon, France: IARC Press; 2002.
- [Google Scholar]
- Clinico-pathological data and prognostic factors in completely resected AJCC stage I-III liposarcomas. Ann Surg Oncol. 2000;7:535-43.
- [Google Scholar]
- Giant primary liposarcoma of the chest. Gen Thorac Cardiovasc Surg. 2009;57:159-61.
- [Google Scholar]
- Soft tissue sarcomas with complex genomic profiles. Virchows Arch. 2010;456:201-17.
- [Google Scholar]
- Pleomorphic malignant fibrous histiocytoma: Fact or fiction. A critical reappraisal based on 159 tumors diagnosed as pleomorphic sarcoma? Am J Surg Pathol. 1992;16:213-28.
- [Google Scholar]
- Pleomorphic liposarcoma: A cytologic study of five cases. Diagn Cytopathol. 2015;43:138-43.
- [Google Scholar]
- Giant retroperitoneal liposarcoma: Report of a case. Ann Urol (Paris). 2002;36:372-5.
- [Google Scholar]
- Immunoreactivity for the human hematopoietic progenitor cell antigen (CD34) in lipomatous tumors. Am J Surg Pathol. 1997;21:195-200.
- [Google Scholar]







