Translate this page into:
Diagnostic relevance of cell block procedure in secondary tumors of the pancreas
*Corresponding author
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Sir,
We have read with great interest the paper entitled “Endoscopic ultrasound-guided fine-needle aspiration diagnosis of secondary tumors involving the pancreas: An institution's experience” by Alomari et al.,[1] which appeared in CytoJournal 2016; 13:1. The authors reported a series of secondary tumors involving the pancreas, either by single or multiple masses, utilizing endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA), also elsewhere considered yet as a safe and reliable diagnostic tool for pancreatic neoplastic lesions.[123] On the other hand, only a few reports have been addressed to study secondary tumors by EUS-FNA involving the pancreas.[14567] We agree the presence of multiple pancreatic or suspicious metastatic lesions elsewhere in the body as well as the occurrence of a prior history of oncological malignancies represent significant clues to suspect the secondary nature of the pancreatic lesion.[1] The key diagnostic dilemma raises when secondary isolated neoplasms exhibit comparable morphological aspects with primary pancreatic carcinomas, mainly when an absent or unknown clinical history of previous neoplasia has been recognized.
Although the efficacy of EUS-FNA partly depends on the size and characteristics of the pancreatic mass as well as on the expertise, training and interaction between endosonographer and cytopathologist, the introduction of cell block procedure (CBP) may be considered an adequate additional diagnostic tool.[123] In fact, this technique allows better preservation of cell architecture with the achievement of routine hematoxylin-eosin staining and finally, the possibility to perform ancillary methods, such as immunohistochemistry or molecular analyses.[2389] In particular, CBP allows the availability of an adequate number of serial sections, with the chance to perform a precise immunohistochemical algorithm for the differential diagnostic purposes; however, also Alomari et al.,[1] stressed the utilization of immunostained cell block sections in selective cases.
We report herein our recent personal unpublished experience concerning three cases of unusual solitary pancreatic solid masses without any documented or reported a history of malignancy elsewhere, obtained by EUS-FNA procedure from three male patients. The specimens were processed by a cytopathologist in-room and immediately examined for adequate cellularity after staining by Diff-Quick and hematoxylin and eosin. This rapid on-site evaluation showed a partially necrotic background with the presence of atypical cells, sometimes as tall columnar cells or arranged in glandular or pseudo-papillary structures, with irregular roundish hyperchromatic dense nuclei, sometimes with evident nucleoli, suggesting thus a diagnosis of positive for malignant cells. Further, the material additionally available was treated for CBP [Figure 1a] and then parallel-serial 5-μm sections were mounted on poly-lysine-coated glasses and submitted to immunocytochemical procedures. To verify the primitive or metastatic nature in each case, a set of the following commercially obtained monoclonal antisera was applied according to manufacturer's recommendations: Cytokeratin 7 (DakoCytomation, Copenhaghen, Denmark; working dilution 1:300), cytokeratin 20 (DakoCytomation; working dilution 1:300), carcinoembryonic antigen (CEA) (DakoCytomation; working dilution 1:500) and CDX2 (DakoCytomation; working dilution 1:50). All of the immunocytochemical reactions were carried out by using a standardized procedure with VENTANA Benchmark automatic immunostainer.
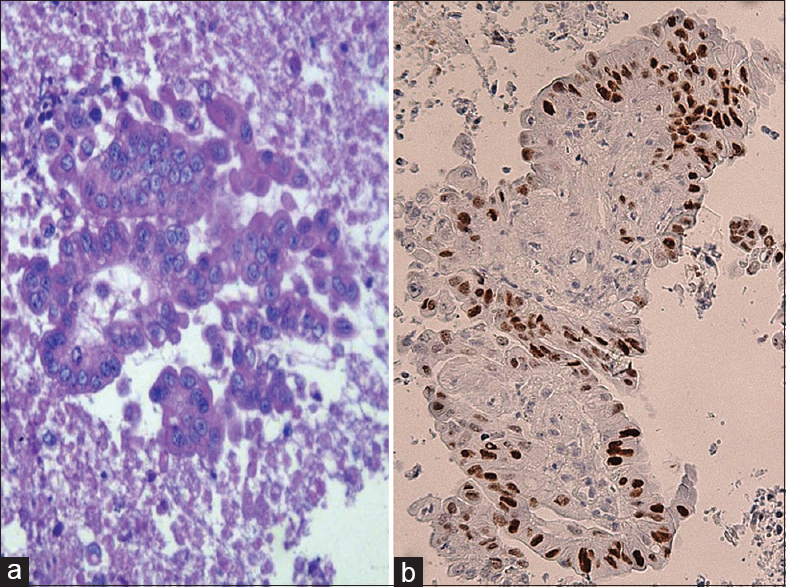
- Cell block procedure sample - pseudoglandular pattern of neoplastic elements from metastatic colonic carcinoma presenting as a pancreatic mass (H and E, ×40) (a). In parallel section, immunohistochemistry revealed a strong nuclear reactivity for CDX2 (Mayer's hemalum counterstain, ×40) (b)
A diffuse and intense cytoplasmic immuno-positivity for cytokeratin 20, CEA and CDX2 was always encountered [Figure 1b], whereas cytokeratin 7 appeared constantly negative. Although CEA staining is not specific and reveals only a few additional support in the differentiation of metastatic colonic from pancreatic ductal adenocarcinoma, the observed immunoprofile was suggestive for a less common metastatic carcinoma coming from the colon, similarly to that reported in cases 16 and 17 by Alomari et al.[1] Consequently, we have investigated to obtain a detailed clinical information regarding the previous occurrence of colonic carcinomas. On this way, the close communication with patient's family and clinicians revealed that two sigmoid and one ascending colon carcinomas had been surgically removed more than 5 years before. Therefore, we fully agree with the suggestion of Alomari et al.,[1] that the cytological examination is an extremely relevant aid to obtain an accurate diagnosis to differentiate the primary or metastatic nature of pancreatic neoplasms, taking into consideration the therapeutic treatment and prognosis should be greatly different to perform.
Finally, the identification and quantification of potential molecular markers may represent a promising field to be further investigated on the same biological samples collected by EUS-FNA CBP assisted since pancreatic metastases from colo-rectal carcinomas consist of a group of heterogeneous disorders with a large number of diverse sets of genetic changes in oncogenes and tumor suppressor genes.
COMPETING INTERESTS STATEMENT BY ALL AUTHORS
The authors declare that they have no competing interests
AUTHORSHIP STATEMENT BY ALL AUTHORS
Each author has participated equally in the work and takes public responsibility for appropriate portions of the content of this article. AI, VB and GT drafted the manuscript and reviewed the literature. All authors have read and approved the nal manuscript.
ETHICS STATEMENT BY ALL AUTHORS
Since this is a manuscript in form of a letter without identifiers, our institution does not require approval from the Institutional Ethical Approval.
LIST OF ABBREVIATIONS (In alphabetic order)
CBP - Cell Block Procedure
CEA - Carcinoembryonic Antigen
EUS-FNA - Endoscopic Ultrasound-Guided Fine-Needle Aspiration.
EDITORIAL/PEER-REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double-blind model (authors are blinded for reviewers and vice versa) through automatic online system.
REFERENCES
- Endoscopic ultrasound-guided fine-needle aspiration diagnosis of secondary tumors involving the pancreas: An institution's experience. Cytojournal. 2016;13:1.
- [Google Scholar]
- Endoscopic ultrasound-guided fine-needle aspiration cytology in pancreaticobiliary carcinomas: Diagnostic efficacy of cell-block immunocytochemistry. Hepatobiliary Pancreat Dis Int. 2015;14:305-12.
- [Google Scholar]
- Cell-block procedure in endoscopic ultrasound-guided-fine-needle-aspiration of gastrointestinal solid neoplastic lesions. World J Gastrointest Endosc. 2015;7:1014-22.
- [Google Scholar]
- Detection of pancreatic metastases by EUS-guided fine-needle aspiration. Gastrointest Endosc. 2001;53:65-70.
- [Google Scholar]
- Diagnosis of nonprimary pancreatic neoplasms by endoscopic ultrasound-guided fine-needle aspiration. Diagn Cytopathol. 2004;31:313-8.
- [Google Scholar]
- Metastatic disease to the pancreas documented by endoscopic ultrasound guided fine-needle aspiration: A seven-year experience. Diagn Cytopathol. 2012;40:228-33.
- [Google Scholar]
- Endoscopic ultrasound-guided biopsy of pancreatic metastases: A large single-center experience. Pancreas. 2013;42:524-30.
- [Google Scholar]
- Gastrointestinal stromal tumours of the stomach: Cytological and immunocytochemical diagnostic features of two cases diagnosed by endoscopic ultrasound-guided fine needle aspiration. Oncol Lett. 2013;5:1862-6.
- [Google Scholar]
- Intraparietal esophageal leiomyomas diagnosed by endoscopic ultrasound-guided fine-needle aspiration cytology: Cytological and immunocytochemical features in two cases. Oncol Lett. 2014;8:123-6.
- [Google Scholar]







