Translate this page into:
Diff-Quik: A reliable stain for rapid onsite evaluation and differentiation of round cells in semen

-
Received: ,
Accepted: ,
How to cite this article: Muniraj F. Diff-Quik: A reliable stain for rapid onsite evaluation and differentiation of round cells in semen. CytoJournal 2022;19:59.
Abstract
Objectives:
Identification of immature germ cells and leukocytes and their differentiation pose difficulty in interpretation. In this study, Diff-Quik (DQ) and Papanicolaou (Pap) are used to differentiate between these two, as they are recommended by the WHO, cost-effective and are easy to use.
Materials and Methods:
This study attempts to differentiate between immature germ cells and leukocytes in semen; compare the usefulness of two stains –Pap and DQ; evaluate the possible associations between the total sperm count and the round cell count; the total sperm count and the presence of round cells and inflammatory cells; the occupation and the sperm concentration; and the occupation and the round cell count.
Results:
A total of 60 semen samples were included in the study. Two smears were made for each patient; DQ stain was used to stain the air-dried smears and Pap stain for alcohol-fixed smears. The whole procedure of DQ staining takes only 30 s. The smears were compared. There was good concordance between DQ and Pap stains in the detection of inflammatory cells and immature germ cells (P = 0.000). There was no association between total sperm count and round cell count (r = −0.086 and P = 0.520); and between the occupation and the sperm concentration or the round cell count.
Conclusion:
There was an inverse relationship between total sperm count and presence of immature germ cells (r = −0.291). There was no association between round cell count and presence of immature germ cells (r = 0.148 and P = 0.269), total sperm count and presence of inflammatory cells, round cell count, and presence of inflammatory cells. DQ stain is easy to use, convenient, and rapid. There is good concordance between DQ and Pap stains in the detection of inflammatory cells and immature germ cells. Total sperm count is independent of the round cell count or the presence of inflammatory cells/immature germ cells. In most of the cases, the round cells were the inflammatory cells. Morphological interpretation can be done even in non-liquefied samples.
Keywords
Cell differentiation
Cytomorphology
Diff Quik stain
Germ cells
Semen analysis
INTRODUCTION
About 30–50% of the couples who have infertility problems have a male-factor as the etiology.[1-4] Semen samples contain two types of cells, one of spermatogenic origin, the other being nonspermatogenic. The spermatogenic cells are the immature germ cells and the mature spermatozoa; the non-spermatogenic cells include inflammatory cells including neutrophils, lymphocytes, macrophages, and the epithelial cells. The reports of semen analysis frequently contain the term “round cells”, without any note about the further specification of the cells. Round cells in semen include immature germ cells, Sertoli cells, and inflammatory cells.[5,6] Cytological interpretation of germ cells is infrequently done in studies on infertility in the form of fine needle aspiration, touch preparation cytology, etc.[4,7,8] Round cells are increasingly evaluated in studies on infertility, as they can denote the specific cause for the infertility.[9] The presence of immature germ cells in semen may be indicative of testicular damage, germ cell maturation arrest.[5,9] Reduced sperm counts are known to be associated with increased numbers of immature germ cells.[6,9] In azoospermic individuals, absence of immature germ cells in semen indicates obstructive cause, whereas the presence of immature germ cells indicates endocrinological disorder. Even with normal sperm counts, the presence of immature germ cells in semen accounts for impaired fertility in males.[2,5] Increased numbers of leukocytes in semen reflect infection/inflammation of the male genital tract including sexually transmitted diseases and poor quality of the sperms. Leukocytes are known to affect the motility of the sperms and thus the fertility.[2,5,6,9,10-12] In the study by De Bellabarba, the highest leukocyte concentration was observed in infertile men, compared to fertile men.[11] Identification of immature germ cells and leukocytes and their differentiation pose difficulty in interpretation.[2,13] Sub-fertility due to leukocytospermia can easily be treated with antibiotics. Hence, the differentiation of round cells as either inflammatory cells or immature germ cells is mandatory. Further, with the recent advanced techniques, immature germ cells from azoospermics can be used for in vitro fertilization and cytogenetic assay.[2] Hence, it is essential to differentiate between the leukocytes and immature germ cells to treat the patient accordingly. Various stains are available to assess the sperm morphology and to differentiate between immature germ cells and leukocytes.[1-6,9,11,13-16] They are:
Myeloperoxidase stain
Leishman stain
Diff-Quik (DQ) stain
Papanicolaou (Pap) stain
Hematoxylin and Eosin
Giemsa
May-Grünwald-Giemsa
Toluidine blue.
Of these, we prefer to use Pap and DQ, as they are recommended by the World Health Organization (WHO), cost-effective and are easy to use.[10] DQ is especially useful because of its simple and rapid staining procedure. Peroxidase is mainly recommended for identifying granulocytes.[10]
Objectives
This study attempts to
Differentiate between immature germ cells and leukocytes in semen;
Compare the usefulness of two stains –Pap and DQ;
Evaluate the possible associations between the total sperm count and the round cell count; between the total sperm count and the presence of round cells and inflammatory cells; between the occupation and the sperm concentration; and that between the occupation and the round cell count.
MATERIAL AND METHODS
This study was cross-sectional and was commenced after getting approval from the Institutional Human Ethics Committee. A total of 60 semen samples, submitted to the clinical pathology laboratory in Chettinad Hospital and Research Institute, for routine investigation of infertility, were included in the study. After getting informed written consent, the details of the patient such as age, occupation, period of abstinence, clinical details of the patient’s spouse, and previous investigations if relevant were collected for every patient. Patient identity and the details of the information collected for the study were kept confidential. The patient was informed about the procedure and informed consent was obtained from each patient. Semen samples in which no round cells were found, samples whose quantity was just sufficient to do the routine semen analysis, and smears which were washed out during processing with either of the stains were excluded from the study. Smears were prepared from the samples which fulfilled the inclusion criteria. The smears were made from the samples, by “feathering method” as recommended by the WHO by placing a drop of the undiluted semen on the slide, spreading it with the help of another slide kept over the drop of the sample at acute angle by pulling it forward.[2,10] The smears were made about 1 h after collection, irrespective of the status of liquefaction, after adequate amount of the sample was taken for routine analysis. The making of smears was technically difficult in case of samples which did not liquefy even after 1 h. Two smears were made for each patient; one was air-dried for at least 5 min and the other was fixed in 80% isopropyl alcohol (v/v) (RANKEM, Avantor Performance Materials India Ltd., Thane, India) for at least 30 min. Diff-Quik stain (Sperm Morphology Kit, CryoLab International, Chennai, India) was used to stain the air-dried smears and Pap stain (Nice Chemical (P) Ltd., Kochi, India) for alcohol-fixed smears. DQ stain kit comprises three solutions, in which the smear is sequentially immersed. One is a fixative which is triarylmethane dye dissolved in methanol, in which the air-dried smear is immersed for 15 s; staining solution 1 which is eosinophilic xanthene, in which the fixed smear is immersed for 10 s; and staining solution 2 which is basophilic thiazine, in which the smear is immersed for 5 s. The whole procedure takes only 30 s and it is the method recommended by the WHO for rapid staining of semen.[10,14] The air-dried smears were fixed in the fixative in the Sperm Morphology Kit, for 15 s; and then immersed sequentially in Stain 1 and Stain 2 in the kit for 10 and 5 s, respectively. The excess solutions were drained at each step, by placing the slides vertically on absorbent paper. During Pap staining, the smears were preferably washed in a container of water rather under running tap water, as the smears tend to get washed off. The smears were examined microscopically and round cells in the sample were differentiated as either germ cells or inflammatory cells. Further categorization of the germ cells was done whenever possible.
The germ cells were identified with the following morphological criteria: [2,4,6-10,13]
Spermatogonia are cells which are smaller than spermatocytes and larger than spermatids, have thin rim of cytoplasm, high N: C ratio, and single nucleus which is round to oval, 6–7 µm in diameter, darkly stained, has evenly distributed finely granular chromatin, and has one or two nucleoli
Primary spermatocytes are larger cells, with large centrally located spherical nucleus, 8–9 µm in diameter, coarse or thread-like/woolly nuclear chromatin, and irregular nuclear outline
Secondary spermatocytes are small round cells, with a central spherical dark nucleus, 6–7 µm in diameter (sometimes two nuclei); bi-nucleation is common
Spermatids are small cells measuring approximately 4–5 µm in diameter with scant cytoplasm, small, dark round to oval nucleus, sometimes two in number; may be confused with lymphocytes
Spermatozoa are cells which have a head, a neck, and a tail
Sertoli cells have indistinct cytoplasmic borders, oval nucleus with smooth outline, vesicular chromatin, and prominent large nucleolus.
The non-spermatogenic cells were identified with the following criteria: [2,5,6]
Neutrophils have segmented nuclei; have a diameter of approximately 14 µm; generally, are smaller than germ cells; cytoplasm appears granular; and nucleus has several lobes connected by small bridges.
Lymphocytes are small cells, whose diameter approximately ranges from 7 to 12 µm. They usually show a small cytoplasmic border which stains bluish-green on Pap stain, whereas that of spermatid stains pink.
Macrophages usually have a diameter of 16–30 µm; stain bluish-grey on Pap stain; sometimes contain cytoplasmic vacuoles which, in turn, may contain degenerated cellular debris; show a kidney-shaped or sometimes round nucleus, which is usually eccentric; the nuclei never appear pyknotic/ degenerated.
Squamous cells are large, polygonal in shape, and have ample cytoplasm, pyknotic, or vesicular nucleus depending on the maturation.
The cytoplasm of the immature germ cells decreases with the maturation of the cells.[4] The neutrophils were differentiated from the germ cells by their smaller size and multilobed nuclei.[2] Degenerated spermatids may be mistaken for neutrophils or lymphocytes. Sometimes, these spermatids have two or more nuclei, which mimics neutrophils, but can easily be distinguished from lymphocytes, which are never multinucleated.[5] The cells which had indented/irregular nuclear outline were designated as macrophages. The cells with round nuclear outline were designated as immature germ cells, except for the lymphocytes. The presence of macrophages was taken as a supportive factor for designating the small round cells as lymphocytes, instead of spermatids.
The liquefaction time, round cell count, sperm concentration (millions/mL), and total sperm count were noted for each sample.
The statistical analyses were done with SPSS software version 26.0. Pearson’s correlation coefficient (r value) was calculated for continuous variables, namely, total sperm count and round cell count; Spearman’s rho correlation (r value) between continuous and discrete variables, namely, total sperm count and presence/absence of inflammatory cells; Kappa score for agreement between two methods, that is, DQ versus Pap.
RESULTS
The age of the patients ranged from 18 to 41 years. The most of the patients were in the age group of 26–35 years.
The number of days of abstinence ranged from 3 to 7 days, which is routinely followed for all cases of semen analysis. For one patient, days of abstinence could not be counted, as he was an unmarried 18-year-old male student.
The patients belonged to various occupational fields such as information technology, business, engineering, teaching, student, and chef [Chart 1]. Relatively, more patients were in the groups of laborers and drivers. However, as the group of “laborers” includes participants having diverse occupations, “drivers” constitute the major group presenting with infertility.
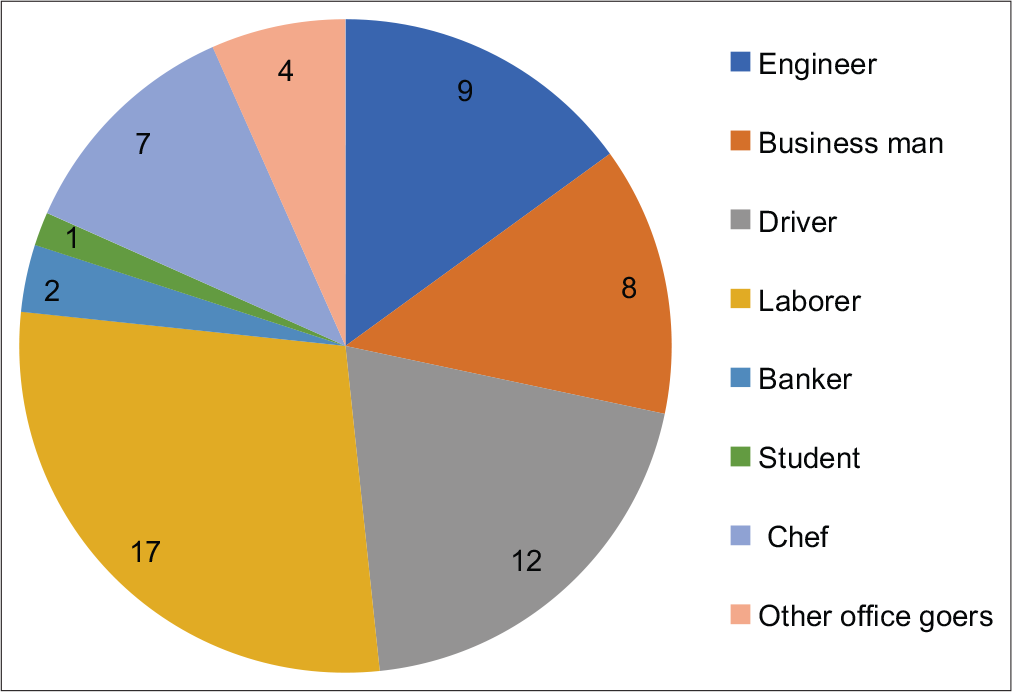
- Occupation of patients
Two of the samples did not liquefy even after 1 h and after repeated syringing. Hence, sperm concentration, total sperm count, and round cell count were not available for these samples. The sperm concentration ranged from 0 to 128 million/mL; the mean concentration was 39.8 million/mL.
The total sperm count ranged from 0 to 330 million/ejaculate; the mean count was 82.6 million/ejaculate.
The round cell count ranged from 0.8 to 13.2 million/mL; the mean count was 3.3 million/mL. Inflammatory cells and no immature germ cells were present in 43 (in DQ, as well as Pap) cases; only immature germ cells and no inflammatory cells were not found in any case; both inflammatory and immature germ cells in 14 cases (in DQ, as well as Pap).
There was good concordance between DQ and Pap stains in the detection of inflammatory cells (r = 0.649) [Chart 2, Table 1a and b] and immature germ cells (r = 1.000) [Chart 3, Table 2a and b] and the correlation was significant in both the cases (P = 0.000).
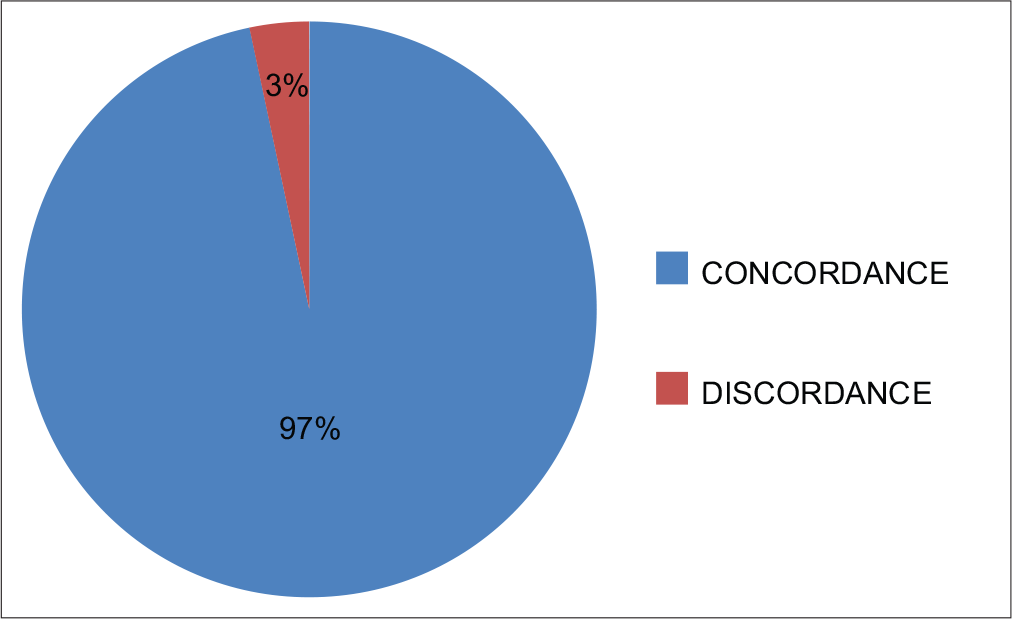
- Concordance between DQ and Pap stain in identification of inflammatory cells
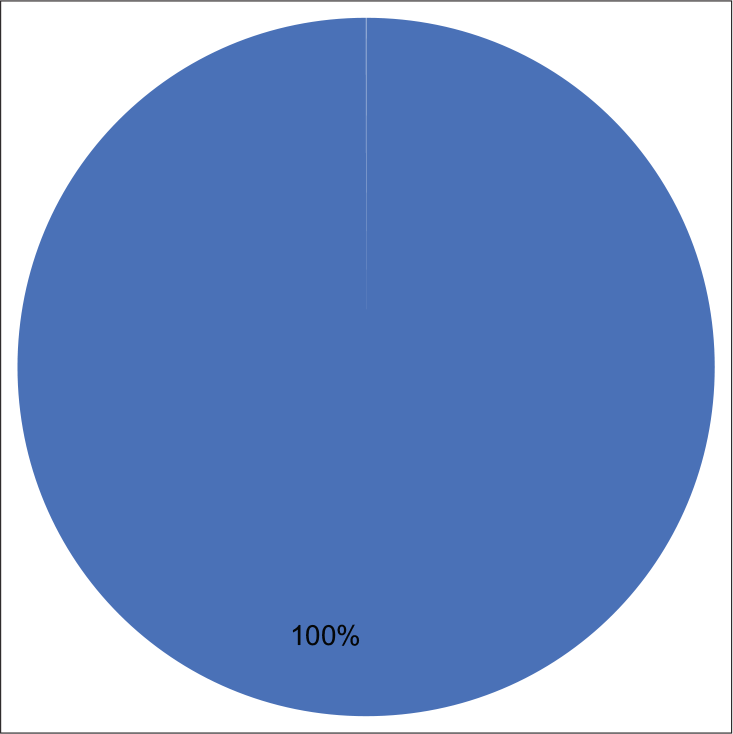
- Concordance between DQ and Pap stains in identification of immature germ cells
| (a) Concordance | ||||
|---|---|---|---|---|
| Inflammatory cells | Pap test | Total | ||
| Absent | Present | |||
| DQ test | ||||
| Absent | 2 | 1 | 3 | |
| Present | 1 | 56 | 57 | |
| Total | 3 | 57 | 60 | |
| (b) Statistical correlation | ||||
| Chi-square value | P-value | |||
| Pearson Chi-square | 25.282 | 0.000 | ||
| Kappa score | 0.649 | 0.000 | ||
| (a) Concordance | |||
|---|---|---|---|
| Immature germ cells | Pap test | Total | |
| Absent | Present | ||
| DQ test | |||
| Absent | 46 | 0 | 46 |
| Present | 0 | 14 | 14 |
| Total | 46 | 14 | 60 |
| (b) Statistical correlation | |||
| Chi-square value | P-value | ||
| Pearson Chi-square | 60.00 | 0.000 | |
| Kappa score | 1.00 | 0.000 | |
There was no association between total sperm count (millions/ejaculate) and round cell count (millions/mL) (r = −0.086 and P = 0.520) [Table 3].
| Round cell count (millions/mL) | Pearson’s/ Spearman’s rho correlation coefficient | |
|---|---|---|
| Total sperm count (millions/ejaculate) | Correlation coefficient (r) | −0.086 |
| P-value | 0.520 |
There was an inverse relationship between total sperm count (millions/ejaculate) and the presence of immature germ cells (r = −0.291) [Table 4, Figure 1a]. This observation was same with both the stains.
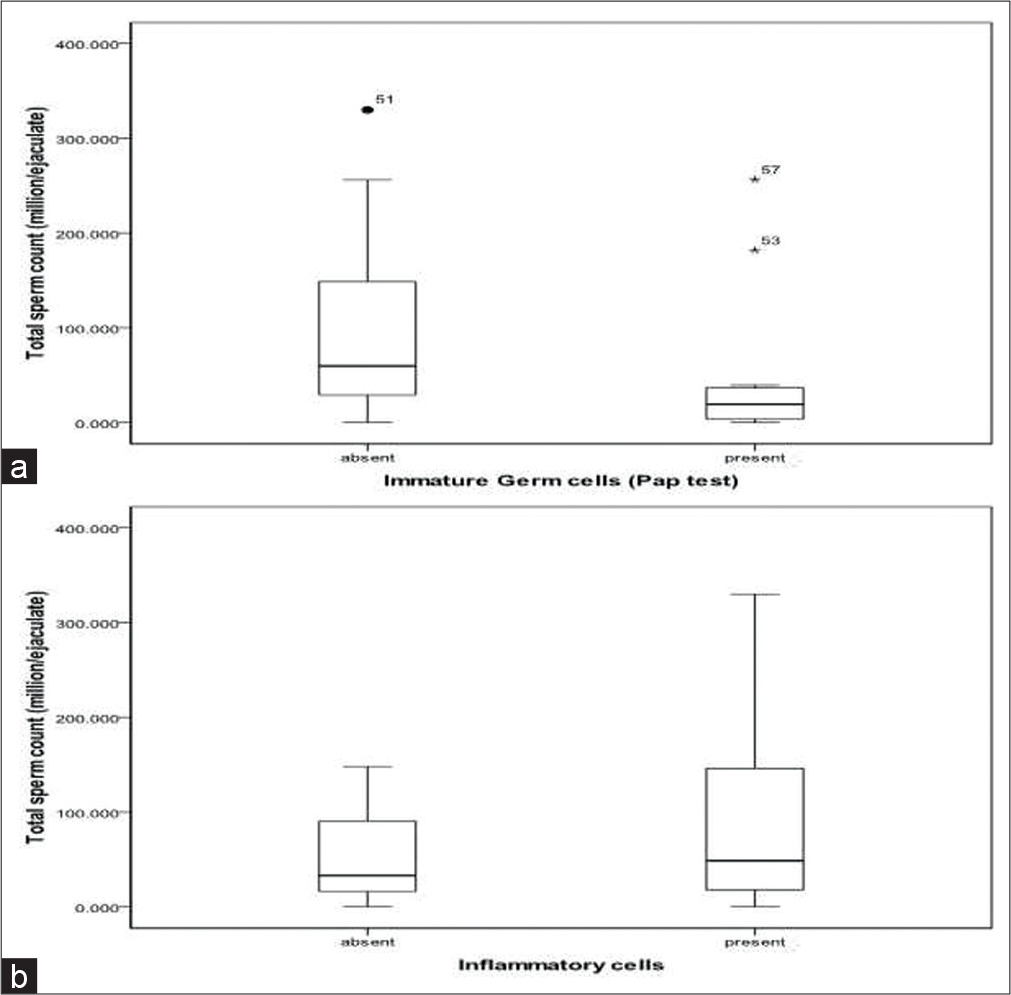
- (a and b) Correlation between the total sperm count and the presence of immature germ cells (a) and inflammatory cells (b).
| Pearson’s/Spearman’s rho correlation co-efficient | Total sperm count (millions/ejaculate) | |
|---|---|---|
| Immature germ cells | ||
| DQ stain | Correlation coefficient (r) | −0.291 |
| P-value | 0.027 | |
| Pap stain | Correlation coefficient (r) | −0.291 |
| P-value | 0.027 | |
There was no association between round cell count and presence of immature germ cells (r = 0.148 and P = 0.269) [Table 5], total sperm count and the presence of inflammatory cells [Table 6, Figure 1b], and round cell count and the presence of inflammatory cells [Table 7].
| Pearson’s/Spearman’s rho correlation co-efficient | Round cell count (millions/mL) | |
|---|---|---|
| Immature germ cells | ||
| DQ stain | Correlation coefficient (r) | 0.148 |
| P-value | 0.269 | |
| Pap stain | Correlation coefficient (r) | 0.148 |
| P-value | 0.269 | |
| Pearson’s/Spearman’s rho correlation co-efficient | Total sperm count (millions/ejaculate) | |
|---|---|---|
| Inflammatory cells | ||
| DQ stain | Correlation coefficient (r) | 0.184 |
| P-value | 0.168 | |
| Pap stain | Correlation coefficient (r) | 0.091 |
| P-value | 0.498 | |
| Pearson’s/Spearman’s rho correlation co-efficient | Round cell count (millions/ml) | |
|---|---|---|
| Inflammatory cells | ||
| DQ stain | Correlation coefficient (r) | −0.123 |
| P-value | 0.356 | |
| Pap stain | Correlation coefficient (r) | −0.049 |
| P-value | 0.716 | |
Out of the two cases of azoospermia, immature germ cells were absent in one and present in the other.
Immature germ cells were present in six, five, and one cases which had normal, reduced and zero sperm concentrations, respectively, and in the two non-liquefied samples.
Mature spermatozoa were readily identified [Figure 2a and b]. Among the immature germ cells, primary spermatocytes [Figure 2c] could be identified relatively easily. Sertoli cells, Spermatogonia, and secondary spermatocytes [Figure 2d] could not be identified with certainty. Spermatids [Figure 2e and f] were confused with lymphocytes. Non-spermatogenic cells that were found were macrophages [Figure 3a and b], neutrophils [Figure 3b and c], lymphocytes [Figure 3d and e], and squamous cells [Figure 3f].
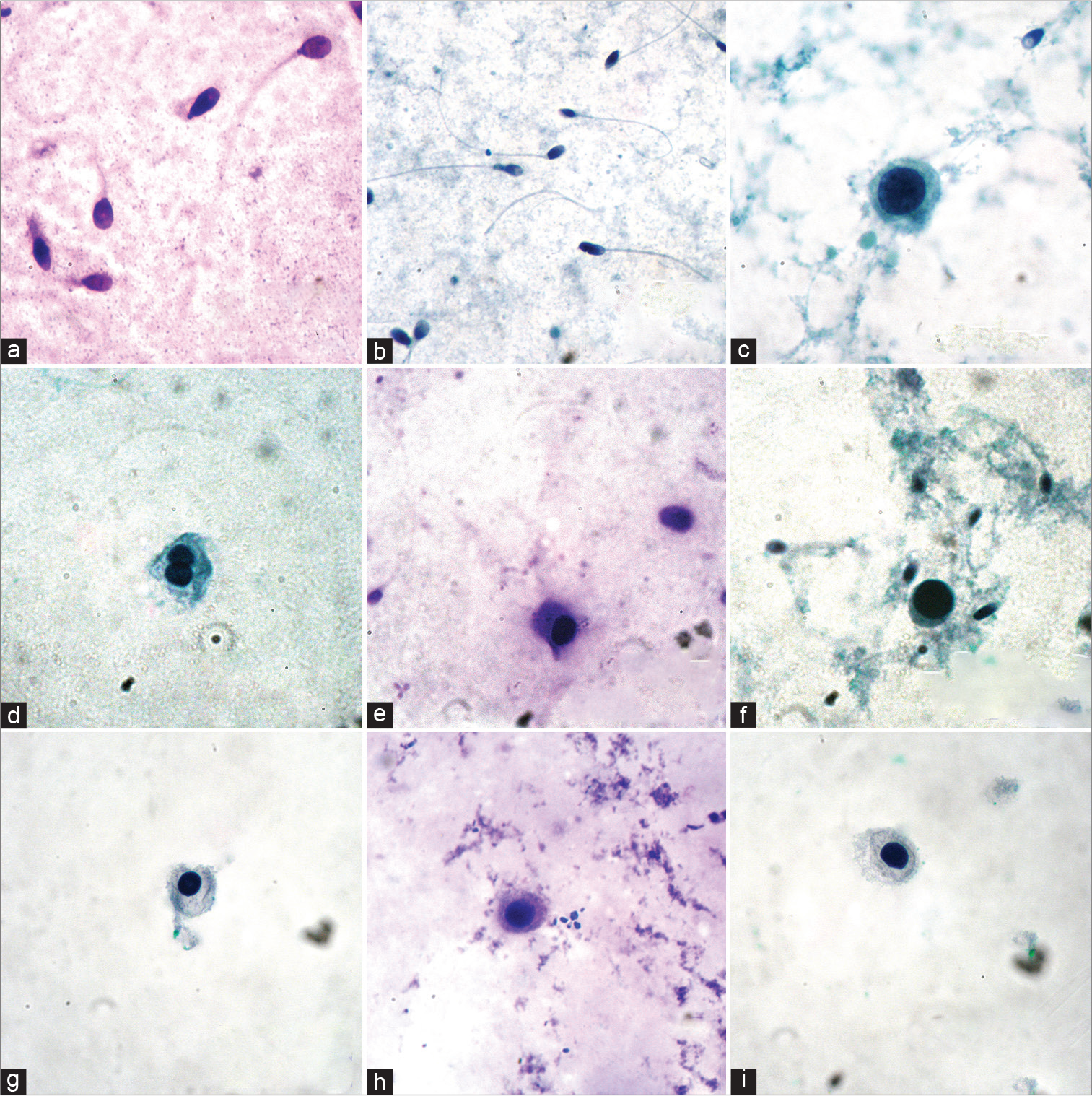
- (a-i) Germ cells in semen: (a) Mature spermatozoa (DQ, ×1000); (b) mature spermatozoa (Pap, ×400); (c) primary spermatocyte (Pap, ×1000); (d) immature germ cell with double nuclei (Spermatid or Secondary Spermatocyte); (e) spermatid (DQ, ×1000); (f) spermatid (Pap, ×1000); (g) immature germ cell (Pap, ×1000); (h) immature germ cell (DQ, ×1000); and (i) immature germ cell (Pap, ×1000).
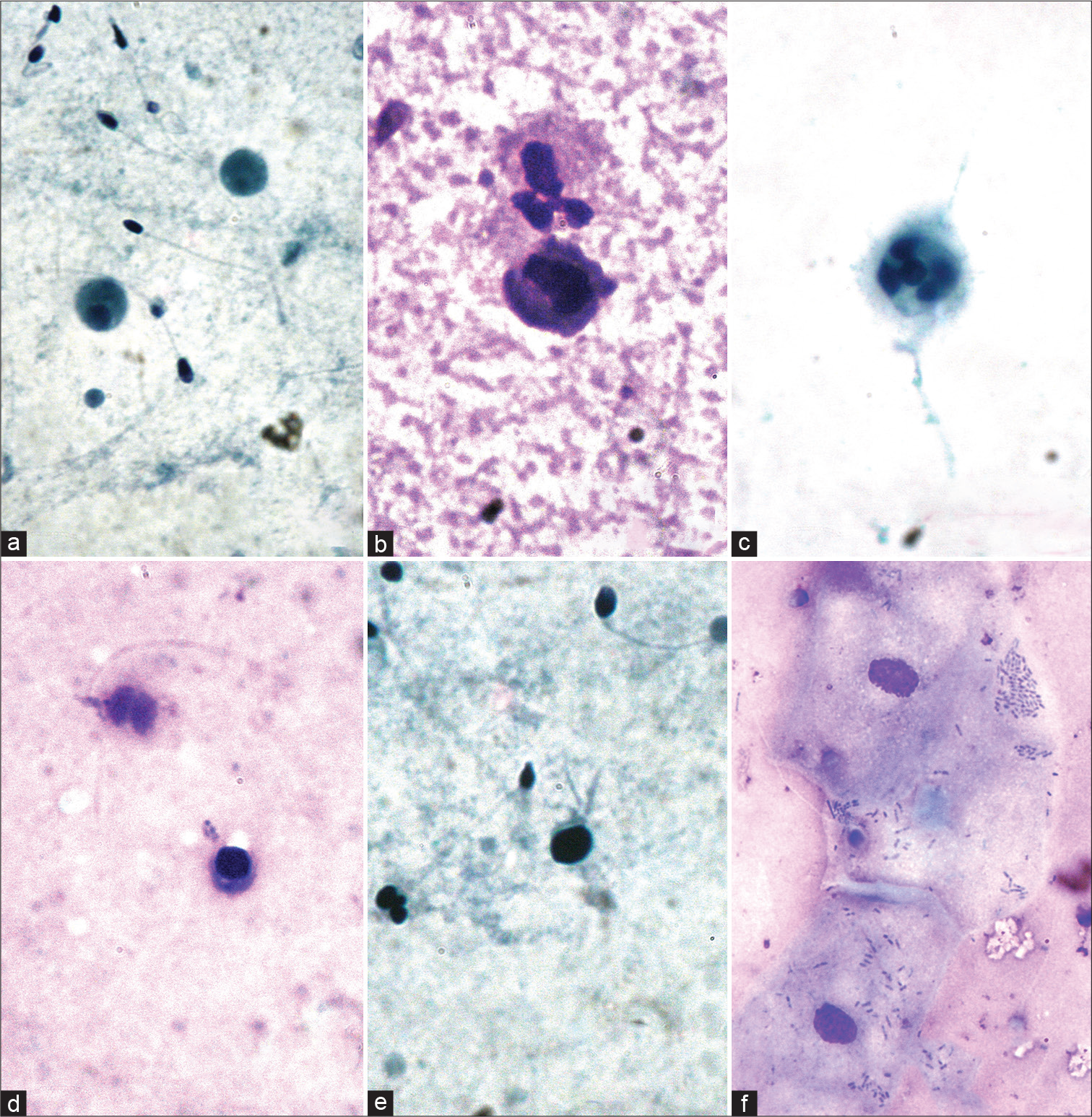
- (a-f) Non-spermatogenic cells in semen: (a) Macrophages (Pap, ×1000); (b) macrophage and neutrophil (DQ, ×1000); (c) neutrophil (Pap, ×1000); (d) lymphocyte (DQ, ×1000); (e) lymphocyte (Pap, ×1000); and (f) squamous epithelial cell (DQ, ×1000).
DISCUSSION
In our study, most of the patients were in the third decade.
In most of the cases, the round cells were inflammatory cells, whereas, in the others, they were immature germ cells [Figure 2g-i]. However, in the study by Williams et al., immature germ cells outnumbered inflammatory cells.[9]
There was no association between the occupation and the sperm concentration [Table 8a] or the round cell count [Table 8b].
| Occupation | Not liquefied | Sperm concentration | ||
|---|---|---|---|---|
| Normozoospermia (>15 million/mL) |
Oligozoospermia (<15 million/mL) |
Azoospermia (0 million/mL) |
||
| Engineer | 1 | 8 | 2 | 0 |
| Business man | 0 | 6 | 0 | 1 |
| Driver | 0 | 10 | 1 | 2 |
| Laborer | 1 | 7 | 6 | 1 |
| Bank sector | 0 | 2 | 0 | 0 |
| Student | 0 | 0 | 1 | 0 |
| Chef | 0 | 5 | 1 | 1 |
| Other office goers | 0 | 3 | 1 | 0 |
| Occupation | Not liquefied | No. of patients with round cell count (>1 million/mL) |
|---|---|---|
| Engineer | 1 | 8 |
| Business man | 0 | 8 |
| Driver | 0 | 11 |
| Laborer | 1 | 14 |
| Bank sector | 0 | 2 |
| Student | 0 | 1 |
| Chef | 0 | 6 |
| Other office goers | 0 | 4 |
Cigarette smoke is not only a carcinogen and mutagen, but it affects the fertility also. In the study by Trummer et al., smokers were found to have more round cell count and the percentage of ejaculates having increased leukocytes were more in case of smokers.[15]
In our study, four patients revealed the history of smoking and they had primary infertility and one patient revealed the history of consumption of alcohol. A patient was an alcoholic.
The other three did not have any other history which could be related to infertility. Three of the four patients had increased round cell count, that is, >1 million/mL. Three (DQ) and two (Pap) of the four patients had leukocytes in semen.
The factors affecting the quality of the semen smear (WHO 2010):[10]
Volume of the semen and the sperm concentration: The degree of overlapping of sperms decreases with the number of sperms
The angle of the spreader: The more acute the angle, the thinner the smear
Smearing speed: The more rapid the movement, the thicker the smear.
Mota and Ramalho-Santos felt that DQ staining is very reproducible, rapid, and simple.[17] The Diff-Quik stain is preferred for its simplicity and rapidity[14] and in our study, we experienced that it was easy to use, did not demand much expertise and convenient as the procedure required less than a minute, and did not require water wash. Pap stain demands more time and expertise which may delay the reporting on morphology.[14] We observed that Pap stain helped to differentiate immature germ cells from leukocytes by highlighting the nuclear outline, which was round in contour in case of immature germ cells, and irregular in case of leukocytes, namely, macrophages, neutrophils, and sometimes in lymphocytes. The condensed chromatin of spermatid was better visualized with Pap stain.
The samples which did not liquefy did not pose any difficulty in the interpretation of the morphology. This is because of the good smearing technique, which distributes the cells evenly [Figure 4].
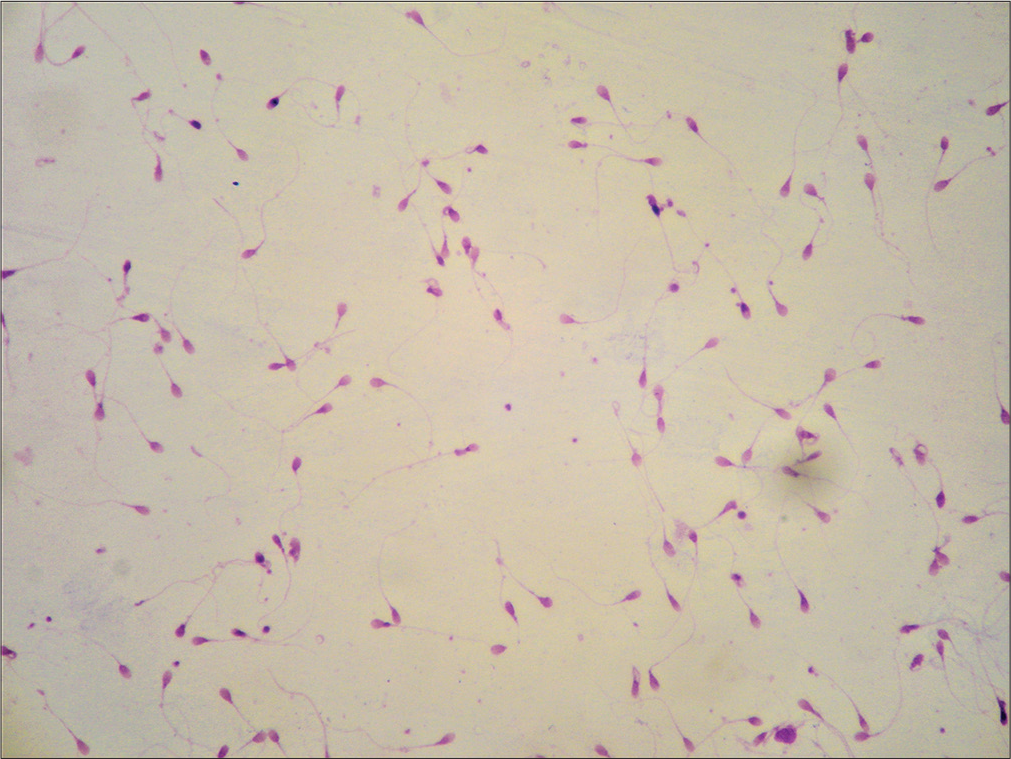
- Well preserved morphology in a non-liquefied sample (DQ, ×100).
In their study on morphological assessment of sperms, Lingappa et al. have stated that the technique including smearing, fixation, and staining should be done meticulously, for the accurate assessment of the sperm morphology. In our study also, morphological assessment and differentiation of the round cells warranted appropriate technical skill at every step of the process.
In the evaluation by Coetzee et al. done with two computer-assisted semen analysis systems, correlation was found to be poorer with Pap stain, compared to DQ stain.[16]
In the study by Patil et al., there was negative correlation between total sperm count and presence of immature germ cells, as is observed in our study [Table 4].
Johanisson et al. have mentioned that some spermatogenic cells and leukocytes are usually present in every semen sample. Various authors have mentioned that differentiation of the round cells into either spermatogenic or leukocytes is sometimes difficult.[5,6]
In our study also, with both the DQ and the Pap stains, primary spermatocytes, though relatively fewer, could be identified relatively easily, compared to other immature germ cells. Fedder has mentioned that primary spermatocytes are fragile, and hence they are broken while preparing the smear.[6] Sertoli cells were not identified in our samples. Cells with double nuclei, probably dividing secondary spermatocytes, were identified. Spermatogonia could not be identified with certainty. Many cells could only be included in the broad category of immature germ cells with these two stains, but could not be categorized further [Figure 2g-i]. Our observation is supported by the study by Williams et al., who, during analysis, had categorized the round cells, into neutrophils, macrophages, lymphocytes, and seminiferous tubule elements for ease of use.[9] Differentiation of spermatids from lymphocytes was difficult. Williams et al. also have observed that differentiation of spermatids from lymphocytes is difficult, as the spermatids are similar in size and shape to lymphocytes.[9] The presence of macrophages was taken as a supportive factor for designating the small cells as lymphocytes, rather than as spermatids. The small cells which had eccentric nuclei were considered as spermatids, rather than as lymphocytes, as is supported by the observation by Foresta and Varotto.[7]
In the study by Patil et al., the immature germ cells that were commonly seen were primary spermatocytes, followed in frequency by spermatids, with occasional presence of binucleate secondary spermatocytes.[2] In our study, spermatids were seen more frequently than the other immature germ cells, similar to the observation by Fedder.[6]
CONCLUSION
Diff-Quik stain is easy to use, convenient, and rapid. Pap stain highlights the nuclear details. Morphological interpretation can be done with both the stains even in nonliquefied samples. There is good concordance between DQ and Pap stains in the detection of inflammatory cells and immature germ cells. Total sperm count is independent of the round cell count or the presence of inflammatory cells/ immature germ cells. This observation was the same with both the stains. In most of the cases, the round cells were the inflammatory cells. The samples which did not liquefy did not pose any difficulty in the interpretation of the morphology. DQ did not demand much expertise, simple, and convenient as the procedure required less than a minute. Pap stain helps to differentiate immature germ cells from leukocytes by highlighting the nuclear outline, which was round in contour in case of immature germ cells, and irregular in case of leukocytes, namely, macrophages, neutrophils, and sometimes in lymphocytes. The condensed chromatin of spermatid was better seen with Pap stain. Morphological assessment and differentiation of the round cells warrant appropriate technical skill at every step of the process.
Acknowledgment
I thank Ms. Helen Monisha, Laboratory Technician, for processing the samples, staining the smears, and helping in analysis.
COMPETING INTEREST STATEMENT BY ALL AUTHORS
The author discloses that she does not have any conflict of interest with respect to this research/publication of this research.
AUTHORSHIP STATEMENT BY ALL AUTHORS
Neither this manuscript nor one with substantially similar content under the author’s authorship has been published or is being considered for publication elsewhere. She has not submitted the same material or portions therefore for consideration of publication in any other journal for which a decision regarding publication is pending. She is aware of the contents of the manuscript and approves this manuscript for submission.
ETHICS STATEMENT BY ALL AUTHORS
The study was started only after getting approval from the Institute Human Ethics Committee. The patients’ details were maintained confidentially. The details of the study including the aims, methods, implications and complications of the study were explained to each participant in the language they could understand and a written informed consent would be obtained prior to sampling.
LIST OF ABBREVIATIONS (IN ALPHABETIC ORDER)
Conc. – Concentration
DQ – Diff-Quik
etc. – etcetera
Hosp. No. – Hospital number
IT – Information Technology
mL – millilitre
Normo – Normozoospermia
N.A. – Not applicable
No. – Number
Oligo – Oligozoospermia
Pap – Papanicolaou
% - Per cent
p-value – Probability value
r – Correlation coefficient
S.A.No. – Semen analysis number
v/v – Concentration of the fluid in volume by volume per cent
WBC – White blood cells
WHO – World Health Organization
EDITORIAL/PEER REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double-blind model (the authors are blinded for reviewers and vice versa) through automatic online system.
References
- Quest for an ideal, simple and cost effective stain for morphological assessment of sperms. J Clin Diagn Res. 2015;9:EC01-4.
- [CrossRef] [PubMed] [Google Scholar]
- Immature germ cells in semen correlation with total sperm count and sperm motility. J Cytol. 2013;30:185-9.
- [CrossRef] [PubMed] [Google Scholar]
- Assessment of spermatozoa morphology under light microscopy with different histologic stains and comparison of morphometric measurements. Int J Morphol. 2012;30:1544-50.
- [CrossRef] [Google Scholar]
- Fine needle aspiration cytology of the testis as the first-line diagnostic modality in azoospermia: A comparative study of cytology and histology. Cytopathology. 2008;19:363-8.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of “round cells” in semen analysis: A comparative study. Hum Reprod Update. 2000;6:404-12.
- [CrossRef] [PubMed] [Google Scholar]
- Nonsperm cells in human semen: With special reference to seminal leukocytes and their possible influence on fertility. Arch Androl. 1996;36:41-65.
- [CrossRef] [PubMed] [Google Scholar]
- Assessment of testicular cytology by fine needle aspiration as a diagnostic parameter in the evaluation of the oligospermic subject. Fertil Steril. 1992;58:1028-33.
- [CrossRef] [PubMed] [Google Scholar]
- Testicular touch preparation cytology in the evaluation of male infertility. Cytojournal. 2011;8:24.
- [CrossRef] [PubMed] [Google Scholar]
- The influence of staining procedure on differential round cell analysis in stained smears of human semen. Biotech Histochem. 1996;71:118-22.
- [CrossRef] [PubMed] [Google Scholar]
- WHO laboratory manual for the examination and processing of human semen (6th ed). Geneva: World Health Organization; 2021.
- [Google Scholar]
- Nonsperm cells in human semen and their relationship with semen parameters. Arch Androl. 2000;45:131-6.
- [CrossRef] [PubMed] [Google Scholar]
- Leukocytic infiltration into the human ejaculate and its association with semen quality, oxidative stress, and sperm function. J Androl. 1994;15:343-52.
- [Google Scholar]
- Immature germ cell separation using a modified discontinuous Percoll gradient technique in human semen. Hum Reprod. 1999;14:1022-7.
- [CrossRef] [PubMed] [Google Scholar]
- A quick, reliable staining technique for human sperm morphology. Arch Androl. 1987;277:275-7.
- [CrossRef] [PubMed] [Google Scholar]
- The impact of cigarette smoking on human semen parameters and hormones. Hum Reprod. 2002;17:1554-9.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of normal sperm morphology outcomes from two different computer-assisted semen analysis systems. Andrologia. 2001;33:159-63.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison between different markers for sperm quality in the cat: Diff-Quik as a simple optical technique to assess changes in the DNA of feline epididymal sperm. Theriogenology. 2006;65:1360-75.
- [CrossRef] [PubMed] [Google Scholar]








