Translate this page into:
Adult rhabdomyoma: A challenging diagnosis on cytology
*Corresponding author
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
To the Editor,
Rhabdomyoma is a rare benign neoplasm of striated muscle, which can be divided into cardiac and extracardiac forms. Of the extracardiac ones, there are three subtypes: adult, fetal, and genital. The adult type has a predilection for the head and neck of elderly males. These tumors originate from the branchial musculature of the third and fourth branchial arches.[1] Clonal structural chromosomal abnormalities including reciprocal translocation between chromosomes 15 and 17 (in the majority) and abnormalities of the long arm of chromosome 10 have been demonstrated in these tumors, indicating that they are neoplasms rather than hamartomas.[2] Given that these tumors are often found in the head and neck area, they lend themselves to fine needle aspiration (FNA).
We would like to present a case of a 63-year-old female who presented to our otolaryngologist with a left-sided hypopharyngeal mass of 5.8 cm. The patient had a diagnosis of granular cell tumor on an FNA from an outside institution. As per the hospital protocol, her outside pathology slides were requested for review. We received FNA slides consisting of Papanicolaou stained smears, H andE stained cytospin preparations, and an Hand stained cell block slide with limited material.
The slides revealed a cellular aspirate composed of large cells with well-defined, abundant, dense eosinophilic cytoplasm and mostly peripherally located nuclei [Figure 1]. The nuclear chromatin was vesicular and the nucleoli were prominent [Figure 2]. There were interspersed blood vessels. No definite cross-striations or crystals were identified. No mitotic activity or necrosis was seen. Immunohistochemical stains could not be performed due to paucity of material. Given the cytomorphology and location of this lesion, the differential diagnosis of adult rhabdomyoma versus granular cell tumor was raised. The tumor was resected and immunohistochemical stains performed on histologic sections revealed the lesion to be diffusely positive for muscle-specific actin, desmin, and myoglobin. The tumor was negative for S100, which supported the diagnosis of adult rhabdomyoma.
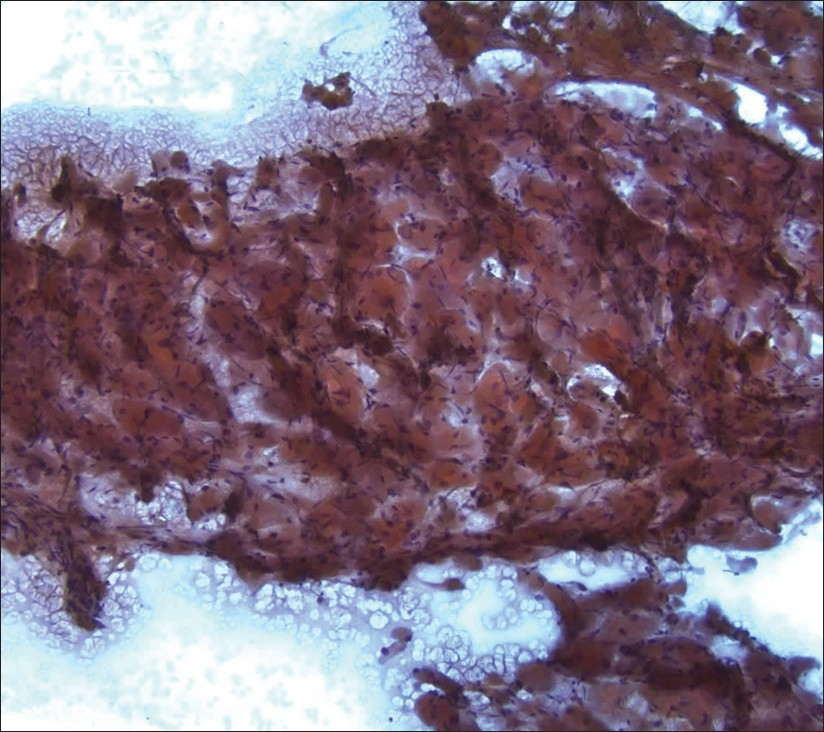
- Adult rhabdomyoma. The lesional tissue had a striking resemblance to skeletal muscle on low-power magnification (Papanicolaou stain, 100×)
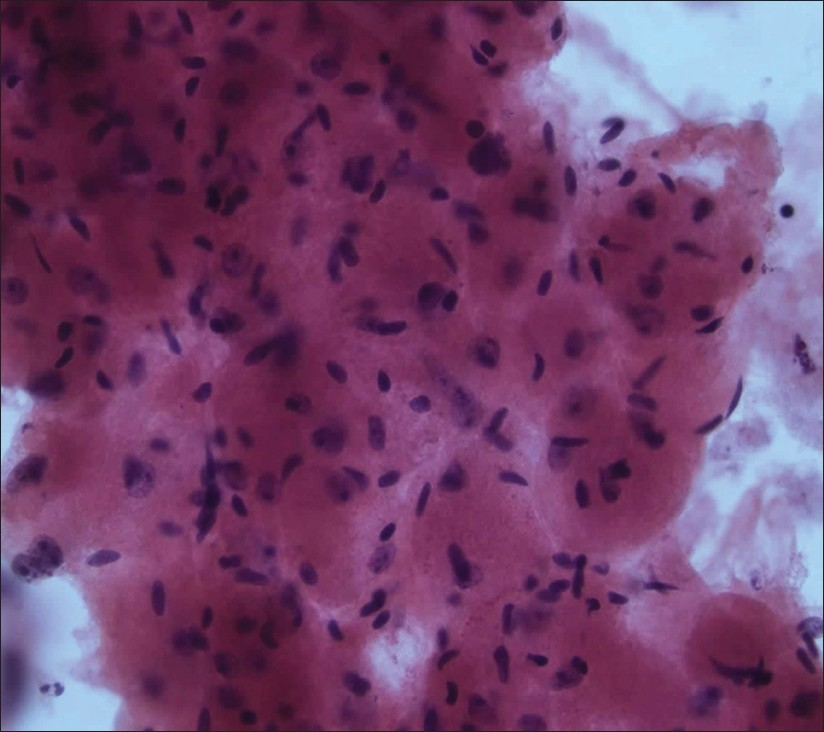
- Adult rhabdomyoma. The cells exhibited abundant eosinophilic dense cytoplasm and peripherally located nuclei with vesicular chromatin and prominent nucleoli (H and E stain, cytospin preparation, 400×)
The cytologic diagnosis of adult rhabdomyoma can be challenging, given the rarity of the tumor and the frequently encountered limited material. It is important to note that many characteristic features of this entity that are readily identified on histology are either missing or scarce on cytology. On histology, the tumor cells are composed of polygonal cells with eosinophilic granular cytoplasm that are intermixed with vacuolated cells. The vacuolation arises due to loss of intracellular glycogen during processing.[3] Further, some of these vacuolated cells appear as spider cells with a central mass of cytoplasm connected via thin strands to a condensed periphery. Striations can be seen in most cases [Figures 3 and 4]. Intracytoplasmic “jackstraw”-like crystalline structures may also be found. In contrast, the cytologic picture lacks the effects of processing. One encounters individually dispersed or large cohesive clusters of round to polygonal cells with abundant eosinophilic granular cytoplasm, peripherally located (at times centrally located) uniform round nuclei with visible nucleoli, and traversing vessels.[3–14] Vacuolated cells have not been described on FNA smears although they have been easily appreciated on formalin-fixed cell block material.[8914] Cross-striations are mostly not identified and, even if present, they are difficult to discern.[3–14] Cytoplasmic crystalline inclusions have only been identified occasionally.[310]
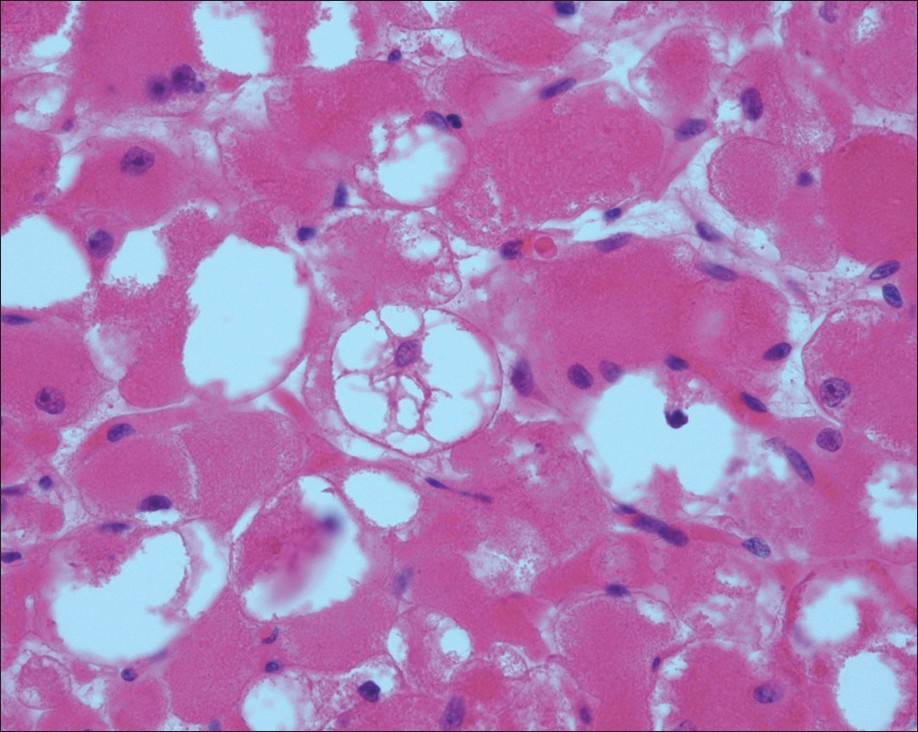
- Adult rhabdomyoma. “Spider cells” as seen on the histologic section of the surgical specimen (H and E stain, 400×)
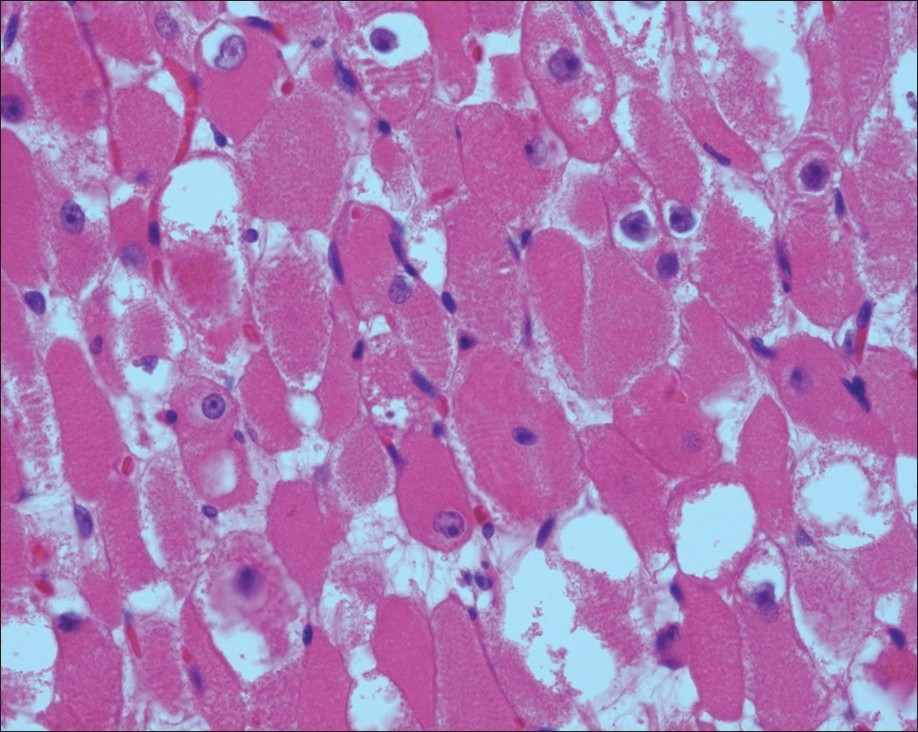
- Adult rhabdomyoma. Striations easily identified on the histologic section of the surgical specimen (H and E stain, 400×)
As is evident from above, lesions harboring cells with abundant granular cytoplasm that are prevalent in the head and neck region should be considered in the differential diagnosis of adult rhabdomyoma. A feature that was very obvious in our case is the remarkable similarity of the neoplastic cells to skeletal muscle on low-power magnification. The cells are large and polygonal with distinct cell borders. However, striations would be easy to find and wellformed in non-neoplastic skeletal muscle. Also, clinical and radiologic information regarding the presence of a well-defined mass would aid in the interpretation. Another entity that can simulate adult rhabdomyoma on cytology is granular cell tumor. These tumors occur in similar locations and have similar polygonal cells with abundant eosinophilic granular cytoplasm in syncytial cell clusters. But the cytoplasm of a granular cell tumor tends to be paler, less sharply defined, more soft and granular, as compared to that of adult rhabdomyoma [Figure 5].[15] A paraganglioma can also be considered in the differential diagnosis with its large polygonal cells, abundant granular cytoplasm, and rich vasculature. But the nuclear chromatin is finely stippled and the nucleoli are indistinct which help in its recognition. Neoplasms including hibernoma (with small cytoplasmic vacuoles), rhabdomyosarcoma (with prominent nuclear atypia), and oncocytoma of salivary gland (smaller cells with centrally located nuclei) can be easily distinguished from adult rhabdomyoma. Recently, a lymphoplasmacytic lymphoma with abundant immunoglobulin inclusions that mimicked adult rhabdomyoma on light microscopy has been described.[16]
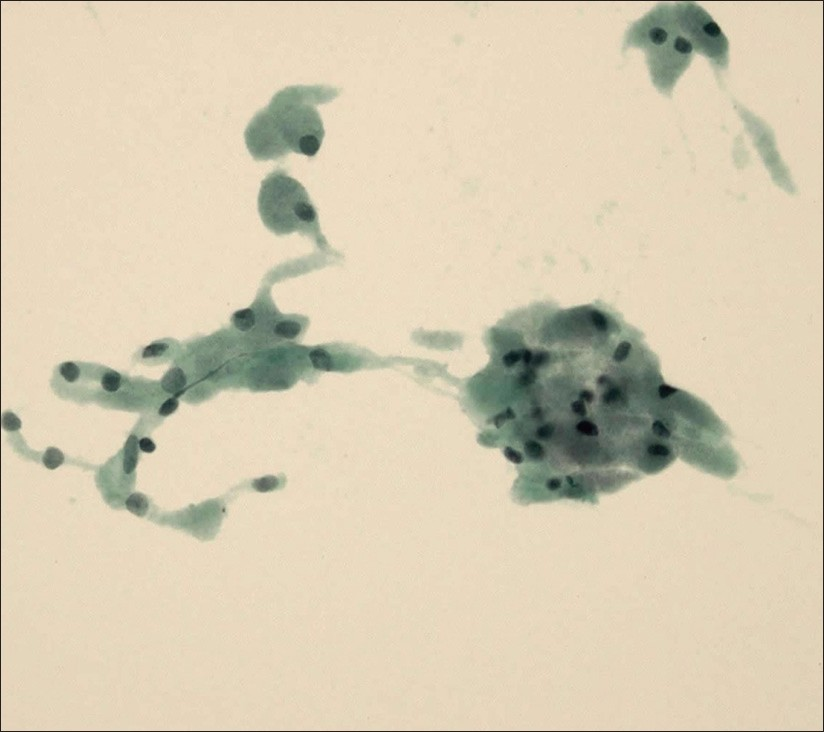
- Granular cell tumor. In contrast to adult rhabdomyoma, the cytoplasm is less well defined, soft, and granular (Papanicolaou stain, ThinPrep, 400×)
Additional material, if available, can be very useful in reaching a definitive diagnosis on FNA alone. The cell block morphology alone may give away the diagnosis with an admixture of granular cells and vacuolated cells and possible presence of striations and crystalline inclusions. Immunohistochemistry can serve as an important adjunct to the diagnosis. Adult rhabdomyomas are positive for desmin, myoglobin, and muscle-specific actin, and negative for S100 and CD68. Granular cell tumors tend to be diffusely positive for S100 and CD68. It is important to note that adult rhabdomyomas may show focal positivity to S100, so more than one stain should be performed when the differential diagnosis of adult rhabdomyoma and granular cell tumor is under consideration.[1] Paragangliomas exhibit S-100 positivity in the sustentacular cells and are also positive for markers of neuroendocrine differentiation such as chromogranin and synaptophysin.
In most cases, adult rhabdomyomas can be treated by local excision. However, recurrences have been described in as many as 42% of cases, usually due to incomplete excision.[17] FNA can be an important tool in making a pre-operative diagnosis of adult rhabdomyoma, thereby reassuring the patient that the tumor is benign and enabling the correct management.
COMPETING INTEREST STATEMENT BY ALL AUTHORS
The authors declare that they have no competing interests.
AUTHORSHIP STATEMENT BY ALL AUTHORS
All authors of this article declare that they qualify for authorship as defined by ICMJE. All authors participated in its design and coordination, and worked collaboratively to draft the manuscript. All authors read and approved the final manuscript.
ETHICS STATEMENT BY ALL AUTHORS
This manuscript is a case description in form of a letter. Therefore IRB approval was not mandatory by our institution.
EDITORIAL / PEER-REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double-blind model (authors are blinded for reviewers and vice versa) through automatic online system.
Available FREE in open access from: http://www.cytojournal.com/text.asp?2012/9/1/20/100124
REFERENCES
- Rhabdomyoma. In: Weiss W, Goldblum JR, eds. Soft Tissue Tumors (5th ed). Philadelphia: Mosby; 2008. p. :584-8.
- [Google Scholar]
- Recurrent parapharyngeal rhabdomyoma. Evidence of neoplastic nature of the tumor from cytogenetic study. Am J Surg Pathol. 1992;16:721-8.
- [Google Scholar]
- Adult-type rhabdomyoma of the palate. Cytologic presentation of two cases with histologic and immunologic study. Acta Cytol. 1990;34:413-9.
- [Google Scholar]
- Pitfalls in the diagnosis of adult rhabdomyoma by fine needle aspiration: report of a case and a brief literature review. Diagn Cytopathol. 2009;37:483-6.
- [Google Scholar]
- Adult rhabdomyoma in fine needle aspirates. A report of two cases. Acta Cytol. 2000;44:223-6.
- [Google Scholar]
- Cytology of a giant adult-type rhabdomyoma of the tongue. Diagn Cytopathol. 2011;39:686-8.
- [Google Scholar]
- Multifocal rhabdomyoma of the neck. Report of a case studied by fine-needle aspiration, light and electron microscopy, histochemistry, and immunohistochemistry. Am J Surg Pathol. 1989;13:791-9.
- [Google Scholar]
- Fine-needle aspiration of adult rhabdomyoma: a case report with review of the literature. Diagn Cytopathol. 2003;28:92-5.
- [Google Scholar]
- Fine needle aspiration cytology of a rhabdomyoma of the pharynx. Acta Cytol. 1986;30:528-32.
- [Google Scholar]
- Fine-needle aspiration cytology of an adult rhabdomyoma of the head and neck. Diagn Cytopathol. 1988;4:152-5.
- [Google Scholar]
- Recurrent multifocal adult rhabdomyoma diagnosed by fine-needle aspiration cytology: report of a case and review of the literature. Diagn Cytopathol. 1990;6:354-8.
- [Google Scholar]
- Diagnosis of adult rhabdomyoma by fine needle aspiration cytology: a report of 2 cases. Acta Cytol. 2010;54(5 Suppl):968-72.
- [Google Scholar]
- Fine needle aspiration of parapharyngeal space adult rhabdomyoma: a case report. Acta Cytol. 2010;54(5 Suppl):775-9.
- [Google Scholar]
- Features of benign granular cell tumor on fine needle aspiration. Acta Cytol. 1999;43:552-7.
- [Google Scholar]
- Primary extranodal soft-tissue B-cell lymphoma with abundant immunoglobulin inclusions mimicking adult rhabdomyoma: a case report. J Med Case Rep. 2011;5:53.
- [Google Scholar]
- Adult rhabdomyoma of the head and neck: a clinicopathologic and immunophenotypic study. Hum Pathol. 1993;24:608-17.
- [Google Scholar]







