Translate this page into:
Fine needle aspiration-induced vascular proliferation of the thyroid: A report of two cases
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
To the Editor,
The most common complication of fine-needle aspiration (FNA) of the thyroid is hematoma formation[1] that rarely causes severe pain due to bleeding into the nodule warranting surgical excision. FNA-induced Masson's vegetant intravascular hemangioendothelioma in a thyroidectomy specimen has brought to light the organizational changes in FNA-induced hematoma.[2] The present report describes two cases of vascular proliferation within the thyroid gland, an organizational change in post-FNA hematoma, with a capillary haemangioma like proliferation in one case.
A 30-year-old female presented to the surgical out-spatient department with a recurrent multinodular goiter. Thyroid function tests were normal. On ultrasonography, both lobes of the thyroid were enlarged. FNA of the thyroid gland showed only hemorrhage. Subsequently, patient underwent near total thyroidectomy. Histopathological examination revealed dense vascular proliferation within the goitrous thyroid gland resembling a capillary hemangioma [Figure 1]. The vascular proliferation was due to organizational changes in a hematoma secondary to the FNA and was wrongly diagnosed as a capillary hemangioma initially.
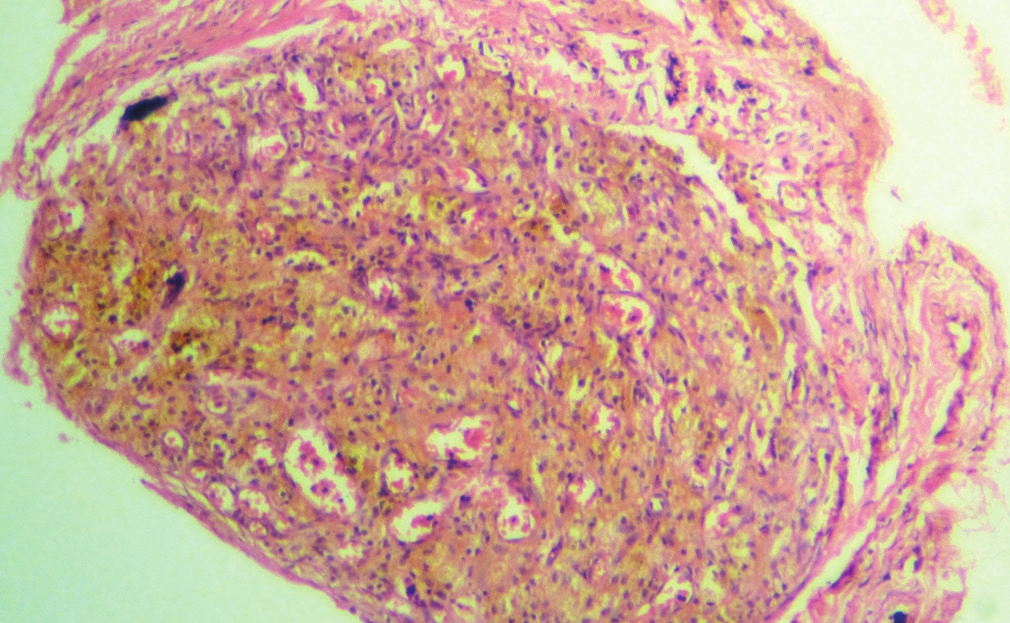
- Vascular proliferation in a hematoma resembling a capillary hemangioma (H and E, ×200)
A 50-year-old female came to the surgical out-patient department with a gradually increasing anterior neck swelling of one year duration. Thyroid function tests were normal. On ultrasonography, there was an ill-defined anechoic area measuring 2.5 × 1.6 cm in the midpole of the left lobe of thyroid, extending into isthmus. The right lobe was normal. There was no cervical lymphadenopathy. A clinical diagnosis of solitary thyroid nodule was given.
On FNA, 3cc of brown fluid was aspirated and the swelling decreased markedly in size. Smears revealed a hemorrhagic background admixed with colloid against which were seen numerous cyst macrophages. A diagnosis of cystic colloid nodule was entertained. Patient underwent partial thyroidectomy. Histopathological examination revealed adenomatous change in colloid goiter with vascular proliferation in hematoma secondary to the FNA [Figure 2].
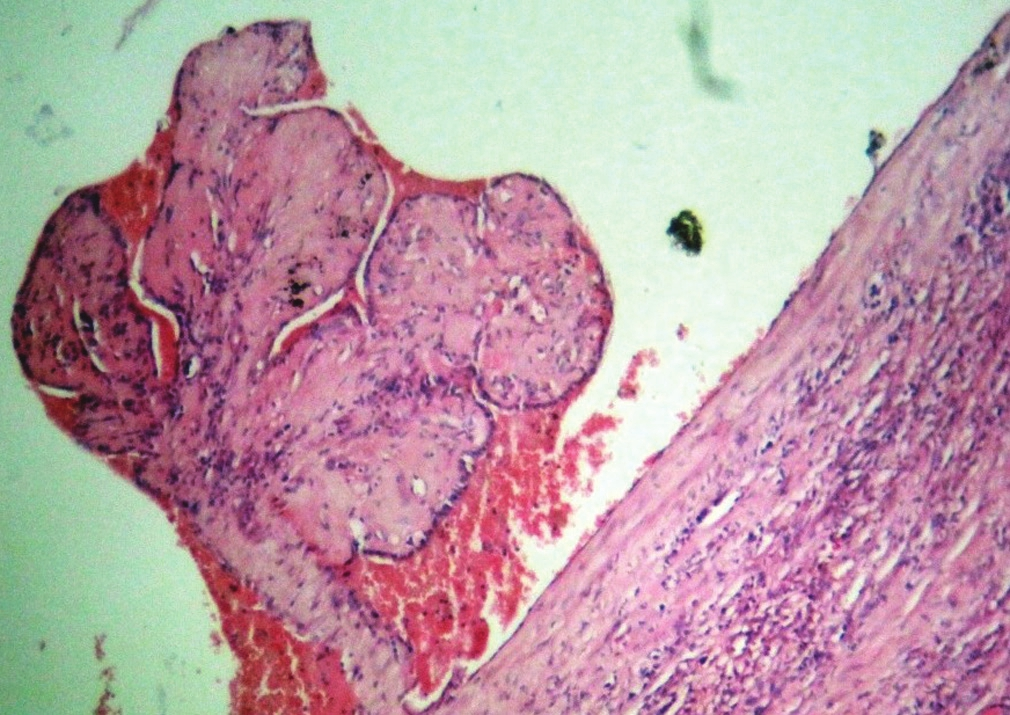
- Organization of a hematoma showing papillae-like structures lined by plump endothelial cells (H and E, ×200)
Thyroid was first studied using FNA in 1930 by Martin and Ellis of the Memorial Hospital for Cancer in New York.[3] Scandinavian workers further established the technique.[45]
At present the thyroid is one of the most commonly FNA sampled organs.
Histological alterations in the thyroid gland after FNA have been studied.[67] Changes caused by FNA include haemorrhage, fibrosis, granulation tissue formation, metaplasia of follicular epithelium, and necrosis, with hematoma formation being the most common.[1] They can occasionally obscure the histology of neoplasms.[89] The features of Masson's vegetant intravascular hemangioendothelioma in a previously aspirated thyroidectomy specimen has brought to light the organizational changes in FNA-induced hematoma.[2] Organization of hematoma results in almost complete resolution with minimal scarring. However, sometimes, unusual vascular and fibroblastic response including Masson's vegetant intravascular hemangioendothelioma resembling an angiosarcoma can occur.[10]
K Tsang and M Duggan, in 1992,[11] described two cases of exuberant vascular proliferations in the thyroid occurring secondary to FNA. Prominent vascularity in the thyroid makes it susceptible to hematoma formation. It can also be due to traumatic injury to the gland from palpation or surgery. But these are less likely because palpation associated changes are described as a thyroiditis.
A few thyroidectomy cases have shown changes suggesting malignancy. These were produced by preoperative FNA and were called as worrisome histologic alterations following FNA of thyroid (WHAFFT) by Livolsi and Merino.[9]
Pandit and Phulpagar in 2001 published their study on WHAFFT lesions.[12] WHAFFT were classified into acute and chronic lesions. Acute lesions comprised of hemorrhage, granulation tissue, siderophages, nuclear atypia, poorly formed granulomas, capsule distortion and infarction, necrosis thrombosis, and recanalization. Chronic lesions were hemorrhage, granulation tissue, siderophages, linear fibrosis, nuclear atypia, vascular changes-hemangioma like, angiosarcoma like papillary endothelial hyperplasia, thrombosis, and recanalisation. Capsular pseudoinvasion, infarction, necrosis, metaplasia, calcification, mitosis, and vascular invasion were the other chronic lesions. Hemorrhage and fibrosis are the most common acute and chronic alterations, respectively.
Worrisome histological alterations such as regenerative nuclear changes, vascular proliferations, metaplasias and capsular pseudo invasion may occur in approximately 10% of thyroid excisions following FNA but awareness of these artifact by histopathologists avoids misdiagnosis. Cytopathologists need to be aware that reaspiration within a few weeks of a previous aspirate can show changes resulting from hematoma formation and healing.
In conclusion, FNA-induced hematomas organize and result in a number of histologic alterations including exuberant vascular proliferative changes. Their knowledge is helpful while evaluating the histologic sections of thyroidectomy specimens. Our report adds to the literature on vascular proliferations in a hematoma developing as a result of thyroid FNA.
COMPETING INTERESTS STATEMENT BY ALL AUTHORS
‘The author(s) declare that they have no competing interests’.
AUTHORSHIP STATEMENT BY ALL AUTHORS
Each author has participated sufficiently in the work to take public responsibility for appropriate portions of the content.
ETHICS STATEMENT BY ALL AUTHORS
As this is case report without identifiers, our institution does not require approval from Institutional Review Board (IRB) (or its equivalent).
EDITORIAL/PEER-REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double blind model (authors are blinded for reviewers and vice versa) through automatic online system.
Available FREE in open access from: http://www.cytojournal.com/text.asp?2012/9/1/25/105118
REFERENCES
- Necrosis of thyroid nodules after fine needle aspiration. Acta Cytol. 1985;29:29-32.
- [Google Scholar]
- Papillary endothelial hyperplasia in the thyroid following fine-needle aspiration. Arch Path Lab Med. 1991;115:240-2.
- [Google Scholar]
- Puncture of goiters for aspiration biopsy.A preliminary report. Acta Med Scand. 1952;144:237-44.
- [Google Scholar]
- Thin-needle biopsy in the diagnosis of thyroid disease. Acta Radiol. 1962;58:321-36.
- [Google Scholar]
- Histologic alterations in the thyroid gland after fine-needle aspiration. Diagn Cytopathol. 1997;16:230-2.
- [Google Scholar]
- Histological alterations following thyroid fine needle biopsy: A systematic review. Diagn Cytopathol. 2009;37:455-65.
- [Google Scholar]
- Effect of fine needle aspiration biopsy on the histology of thyroid neoplasms. Acta Cytol. 1993;37:651-4.
- [Google Scholar]
- Worrisome histologic alterations following fine needle aspiration of the thyroid gland (WHAFFT) Pathol Annu. 1994;29:99-120.
- [Google Scholar]
- Benign tumors and tumor like lesions of blood vessels. In: Weiss SW, Goldblum JR, eds. Enzinger and Weiss's Soft Tissue Tumors. Missouri: Mosby St Louis; 2001. p. :877-83.
- [Google Scholar]
- Vascular proliferation of the thyroid. A complication of fine needle aspiration. Arch Pathol Lab Med. 1992;116:1040-2.
- [Google Scholar]
- Worrisome histologic alterations following fine needle aspiration of the thyroid. Acta Cytol. 2001;45:173-9.
- [Google Scholar]







