Translate this page into:
Parietal swelling in an old female: A diagnostic conundrum

*Corresponding author: Swasti Jain, Department of Pathology, ABVIMS and Dr. RML Hospital, New Delhi, India. jain.swasti7@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Jain S, Kaushal M, Ahuja A, Kumar V. Parietal swelling in an old female: A diagnostic conundrum. CytoJournal 2021;18:11.
HTML of this article is available FREE at: https://dx.doi.org/10.25259/Cytojournal_35_2020
A 60-year-old female presented with a swelling on the left side of scalp for the past 10 months. The swelling was gradually progressing in size and was associated with on and off pain. There was no history of neurological deficits, however, there was a history of chest pain for the past 1 month. On examination, there was a 4 × 3 cm bony hard swelling over the left parietal region of scalp. Her vital signs were stable and fundus examination was normal. Neurological examination did not reveal any signs of raised intracranial pressure, cranial nerve palsy, or any neurological deficit. Her routine hematological and biochemical investigations were within normal range except for an elevated erythrocyte sedimentation rate of 60 mm/h. X-ray of the skull showed a well-circumscribed lytic lesion in the left parietal convexity.
Fine-needle aspiration (FNA) from the swelling on the scalp was performed [Figure 1].
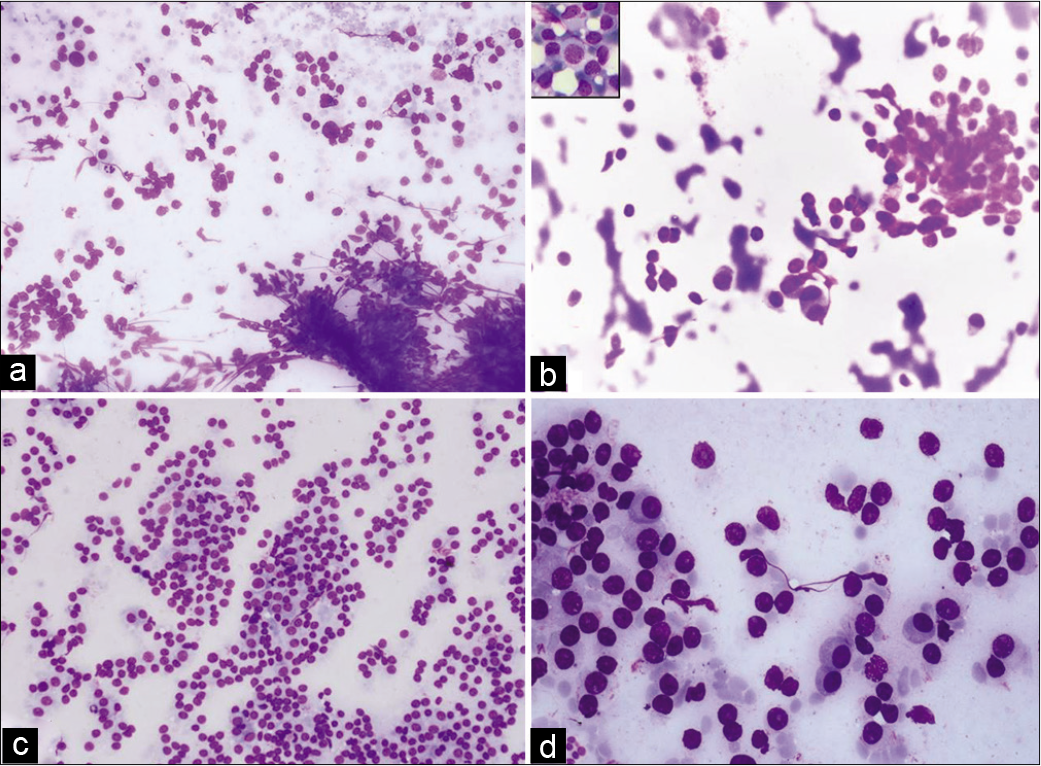
- (a) FNA smears of scalp swelling showing clusters and dispersed round cells (Giemsa, ×100). (b) FNA smears of scalp swelling showing uni- and binucleate cells with eccentric nuclei (Giemsa, ×400), inset: Plasma blast-like cell with dispersed chromatin and moderate cytoplasm. (c) Squash cytology smears of parietal mass showing sheets and scattered oval cells (Giemsa, ×100). (d) Squash cytology smears of parietal mass showing plasmacytoid cells with abundant cytoplasm and eccentrically placed nuclei (Pap, ×400).
Q1. What is your interpretation?
Plasma cell lesion
Hematopoietic cells
Infective etiology
Benign neoplasm
Answer 1: Option a – Plasma cell lesion
Explanation
The aspirate showed moderate cellularity with cohesive fragments of predominantly round cells having inconspicuous nucleoli and fine granular chromatin. Few interspersed cells with eccentric nuclei and pale cytoplasm were noted. Magnetic resonance imaging (MRI) of brain showed an extra-axial homogenously enhancing solid lesion measuring 3.8 × 3.6 cm in the left parietal region involving skull vault and scalp, isointense on T1W [Figure 2a], and heterogeneously hyperintense on T1W with gadolinium [Figure 2b]. There was mild compression of the adjacent brain parenchyma. Rest of the cerebral parenchyma appeared normal with maintained gray-white matter differentiation. Both the cerebellar hemispheres and brainstem showed normal morphology.
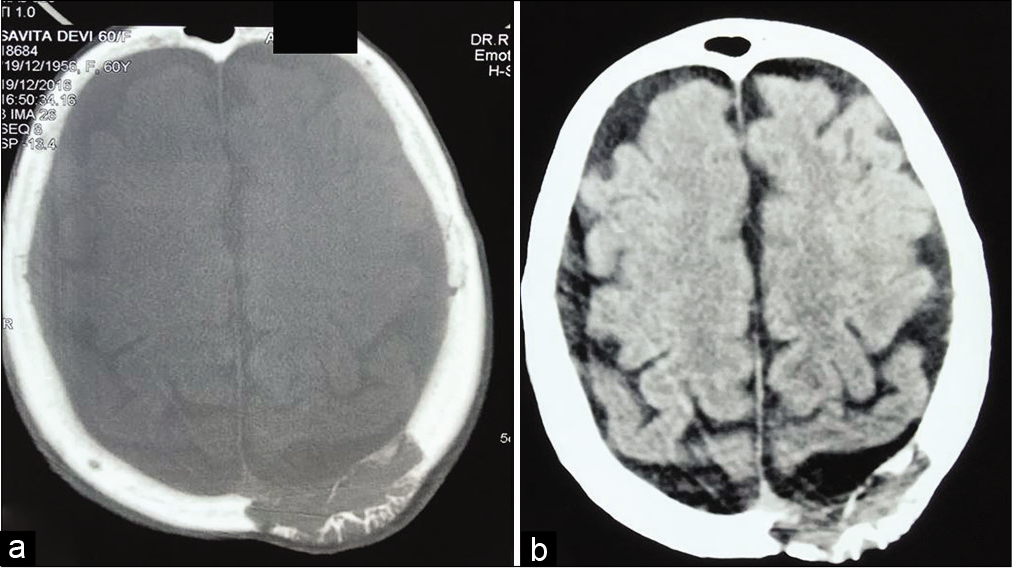
- (a) T1W MRI of the brain showing a well-defined, extra-axial, isointense lesion in the left parietal region. (b) The same lesion is heterogeneously hyperintense on T1W gadolinium-enhanced MRI.
Q2. What is the most likely cytological diagnosis?
Meningioma
Solitary plasmacytoma of skull
Multiple myeloma with associated bony plasmacytoma
Metastatic tumor
Answer 2: Option b – Solitary plasmacytoma of skull
Explanation
Multiple FNA was done from the scalp mass. Smears revealed moderate cellularity comprising clusters of round to oval cells with fine chromatin and inconspicuous nucleoli and also seen were interspersed plasma cells with eccentric nuclei and moderate amount of pale bluish cytoplasm. Binucleate cells with few reactive forms were noted [Figure 1b]. Occasionally, plasmablasts-like cells were seen with high N:C ratio, large round to ovoid, central to eccentric nucleus, and evident nucleoli. The patient was operated for the swelling and intraoperative squash smears were prepared. Smears showed moderate cellularity with sheets and singly dispersed cells of similar morphology with many bi/multinucleate forms [Figure 1c and d]. No evidence of mitosis or necrosis was seen.
Cytomorphological features and radiological findings were suggestive of solitary plasmacytoma of skull. However, a possibility of multiple myeloma (MM) could not be excluded because biochemical investigations were within normal limits, but other ancillary investigations such as complete skeletal survey, serum/urine electrophoresis, and bone marrow examination were not performed.
The diagnosis of meningioma is eliminated as the classical features such as intranuclear cytoplasmic inclusions, whorling pattern, and psammomatous calcification were not found. Sometimes, in rare cases, unusual cytomorphologic features including plasmacytoid appearance may be seen.[1]
Of the metastatic tumors, plasmacytoma may be misdiagnosed as malignant melanoma because of the eccentric nuclei, but it is a less likely diagnosis in our case as there is no pseudo-inclusion, melanin pigment, and frequent mitoses. Metastasis from neuroendocrine tumors (NETs) could still be kept as a differential, however, NET cells are rounder with salt and pepper chromatin, and frequent mitotic figures.
Q3. What is the definite diagnosis?
Solitary plasmacytoma of bone (SBP)
Multiple myeloma with associated plasmacytoma
Meningioma with plasmacytoid pattern
Metastasis
Answer 3: Option b – Multiple myeloma with associated plasmacytoma
Explanation
Surgery for excision of the parietal bony mass lesion was performed. There was grayish-colored tumor tissue adherent to dura which was scraped off. Both the soft tumor tissue and involved bony tissue were sent for histopathological examination. Routine processing was done and hematoxylin and eosin staining was performed. Sections showed sheets of plasmacytoid cells infiltrating the bone [Figure 3a]. Occasional binucleated plasma cells were also noted. The cells strongly expressed CD138 and kappa light chain restriction [Figures 3b-d] and were negative for CD56 and cytokeratin, consistent with the diagnosis of plasmacytoma. The absence of CD56 rules out the possibility of metastatic NET.
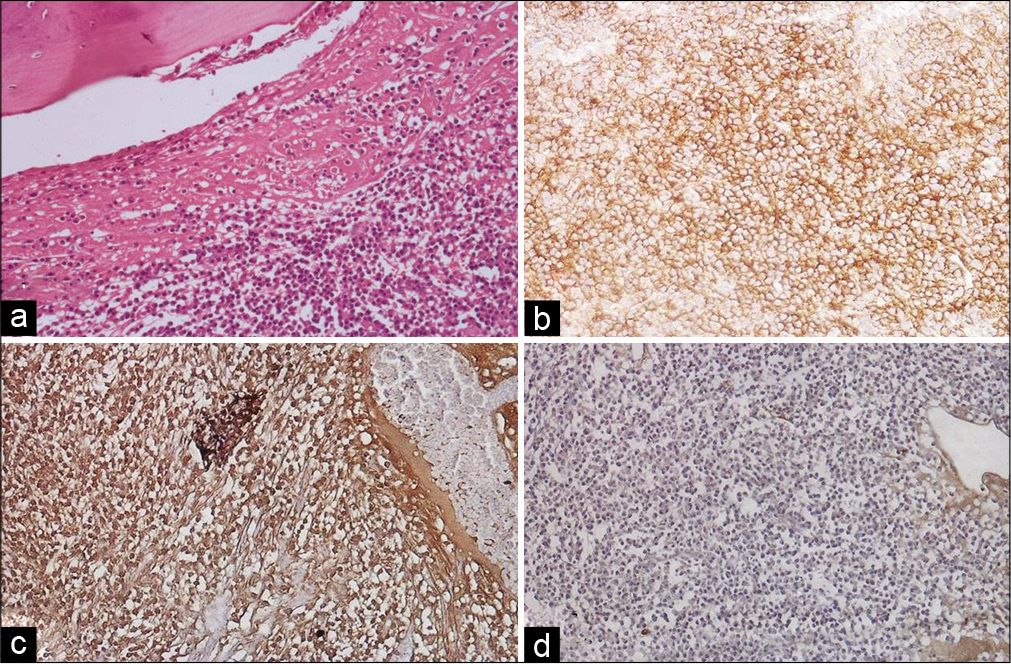
- (a) Tissue sections from parietal mass showing sheets of plasma cells involving the bone (H&E, ×100). (b) Plasma cells showing diffuse membranous positivity for CD 138 (×100). (c) Nuclei of plasma cells showing diffuse kappa positivity (×100). (d) Nuclei of plasma cells negative for lambda (×100).
A workup for MM was advised including all the necessary investigations. Contrast-enhanced computed tomography (CECT) chest revealed multiple erosive lesions in dorsolumbar vertebrae and bony thoracic cage. Serum protein electrophoresis showed monoclonal gammopathy with a serum M protein concentration of 3.50 g/dl [Figure 4]. Serum calcium was 10.1 mg/dl, serum creatinine was 0.6 mg/dl, serum albumin was 3.5 g/dl, and beta-2 microglobulin was 4.2 mg/ml. These findings were consistent with multiple myeloma ISS Stage II. Bone marrow aspiration and biopsy were also performed simultaneously which showed 45% plasma cells with strong CD 38 positivity and kappa restriction. In view of above histomorphological and immunohistochemical (IHC) features, in conjunction with serum electrophoretic and bone marrow findings, a final diagnosis of multiple myeloma with associated plasmacytoma of bone was rendered.
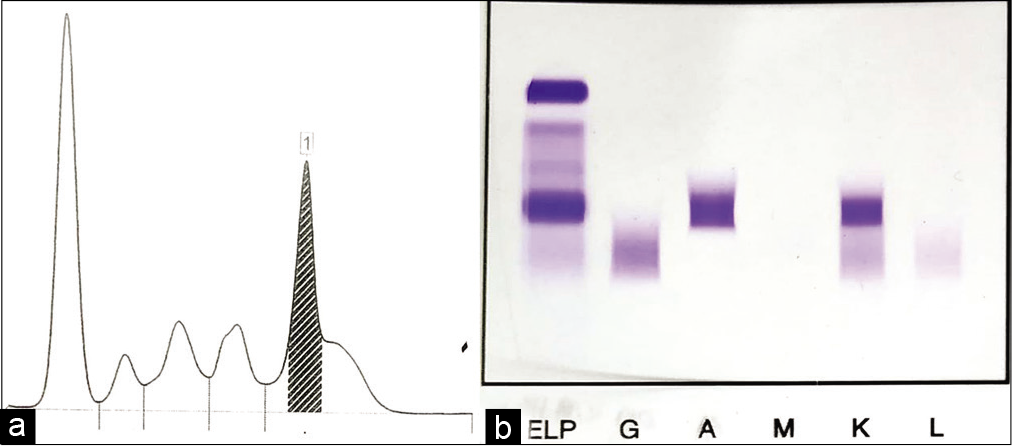
- (a) Serum protein electrophoresis: Densitometric tracing showing M-spike. (b) Serum protein electrophoresis: Gel picture showing M-band.
ADDITIONAL QUIZ QUESTIONS
Q4. What are the symptoms of plasmacytoma of skull?
Bone pain
Bone pain and infection
Cranial nerve palsies
All of the above
Answer 4: Option d – All of the above
Explanation
MM classically presents with pain related to skeletal lytic lesions, anemia, infections, renal failure, and hyperviscosity syndrome. Plasmacytoma of skull can lead to bone pain as well as cranial nerve compression secondary to skull base involvement, mostly affected are II, III, IV, and VI.[2]
Q5: Which of the following statements is correct?
The potential for malignant systemic progression (MM) is higher for SBP than extramedullary plasmacytoma (EMP).
The potential for malignant systemic progression (MM) is higher for EMP than SBP.
The potential for malignant systemic progression (MM) is similar for EMP and SBP.
None of the above statement is correct.
Answer 5: Option a - The potential for malignant systemic progression (MM) is higher for SBP than EMP.
Explanation
It has been found that the risk for a plasmacytoma of skull base to progress to MM is higher in comparison to EMP.[3]
Brief review of the topic
Plasma cell neoplasms arise due to pathologic proliferation of the plasma cell population and the presentation can vary from benign solitary plasmacytoma to malignant MM. Solitary plasmacytoma can either involve an isolated bone or can be seen in the form of EMP. MM usually presents with symptoms secondary to anemia, renal failure, bony lytic lesions, and infections due to hypogammaglobulinemia. The most common location of EMP is head and neck.[4] Cranial plasmacytomas may arise from the skull, leptomeninges, or the brain parenchyma.[5] Calvarium is more common than the base, in case of skull which is similar to the present case.[6] These can be mistaken for meningioma, metastases, or lymphoma radiologically as well as morphologically if it is not associated with MM. Meningioma is the most common mimicker in such cases.[5] It is possible for an apparent solitary plasmacytoma to be the first or even the sole manifestation of underlying MM.[6] The incidence of concomitant EMP at the diagnosis of MM is about 7–18%.[7] In our case, it is hard to comment whether plasmacytoma had gradually transformed into MM or it was the initial presentation of MM.
Meningiomas are the most common extra-axial tumors which present with headache and focal neurological deficits depending on the location. A few case reports of plasmacytoma mimicking meningioma on radiology have been found. Meningiomas are usually isointense on T1W imaging and show post-contrast enhancement which is similar to plasmacytomas. Calcification can be seen in about 20% of cases.[8] The morphology has the characteristic meningothelial whorls, intranuclear inclusions, and psammoma bodies.[1] The unusual plasmacytoid appearance can lead to a misdiagnosis and can be ruled out by IHC which shows positivity for vimentin, epithelial membrane antigen (EMA), and S100.
Majority of the dural metastatic tumors originate from breast, lung, kidney, prostate, lymphoma, or a melanoma.[9] Like meningiomas, they are isointense on T1W and exhibit intense contrast enhancement.[8] Positron emission tomography-computed tomography (PET/CT) scan and the biopsy of the lesion prove a great help in these cases.
Primary dural lymphomas are the rare malignancies in the central nervous system (CNS) and are usually low-grade marginal zone associated lymphomas. These are again isointense on T1W and show vivid post-contrast enhancement.[8] Secondary lymphomas to CNS are usually B-cell type, most commonly diffuse large B-cell lymphoma followed by Burkitt lymphoma.[10] Hodgkin lymphoma only rarely metastasizes to brain.
The treatment modality is different in different types of plasma cell neoplasms. Excisional surgery is the treatment of choice in case of solitary plasmacytoma. However, radiotherapy may be indicated in some cases depending on the location and the extent of the tumor.[11] Chemotherapy has no role until there is systemic involvement by MM.
SUMMARY
Solitary plasmacytoma can be the initial presentation of multiple myeloma so a thorough clinical, hematological, and biochemical workup along with skeletal survey is necessary in all cases. Correct diagnosis in such cases helps in planning proper treatment and results in prolonged overall and progression-free survival.
COMPETING INTEREST STATEMENT BY ALL AUTHORS
The authors declared that they have no competing interest.
AUTHORSHIP STATEMENT BY ALL AUTHORS
Each author has participated sufficiently in the work and takes public responsibility for appropriate portions of the content of this article. All authors read and approved the final manuscript. Each author acknowledges that the final version was read and approved.
ETHICS STATEMENT BY ALL AUTHORS
As this is case without identifiers, our institution does not require approval from the Institutional Review Board (or its equivalent).
LIST OF ABBREVIATIONS (In alphabetic order)
CECT – Contrast-enhanced computed tomography
CNS – Central nervous system
EMP – Extramedullary plasmacytoma
FNAC – Fine needle aspiration cytology
MM – Multiple myeloma
MRI – Magnetic resonance imaging
NET – Neuroendocrine tumor
SBP – Solitary plasmacytoma of bone.
EDITORIAL/PEER-REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double-blind model (authors are blinded for reviewers and vice versa) through automatic online system .
References
- Extradural spinal meningioma as a source of plasmacytoid cells. A case report. Acta Cytol. 2007;51:68-72.
- [CrossRef] [PubMed] [Google Scholar]
- Skull base presentation of multiple myeloma. ENT J. 2011;90:6-9.
- [CrossRef] [PubMed] [Google Scholar]
- Skull base plasmacytoma in a patient with light chain myeloma. Skull Base. 2003;13:167-71.
- [CrossRef] [PubMed] [Google Scholar]
- Guidelines on the diagnosis and management of solitary plasmacytoma of bone and solitary extramedullary plasmacytoma. Br J Haematol. 2004;124:717-26.
- [CrossRef] [PubMed] [Google Scholar]
- Calvarial plasmacytoma mimicking meningioma as the initial presentation of multiple myeloma. Cureus. 2017;9:e1126.
- [CrossRef] [PubMed] [Google Scholar]
- Plasmacytoma of the base of skull-a case report. Ind J Med Paediatr Oncol. 2007;28:34-7.
- [Google Scholar]
- Soft-tissue plasmacytomas in multiple myeloma: Incidence, mechanisms of extramedullary spread, and treatment approach. J Clin Oncol. 2011;29:3805-12.
- [CrossRef] [PubMed] [Google Scholar]
- Multiple myeloma presenting as dural plasmacytoma. Radiol Case Rep. 2019;14:1007-13.
- [CrossRef] [PubMed] [Google Scholar]
- Dural based mass: Malignant or benign. J Radiol Case Rep. 2009;3:1-12.
- [CrossRef] [PubMed] [Google Scholar]
- Secondary central nervous system lymphoma: Spectrum of morphological MRI appearances. Neuropsychiatr Dis Treat. 2018;14:733-40.
- [CrossRef] [PubMed] [Google Scholar]
- Surgical treatment for a giant solitary plasmacytoma with skull erosion. Cureus. 2018;10:e3535.
- [CrossRef] [Google Scholar]







