Translate this page into:
Primary cutaneous non-Hodgkin's lymphoma, clinically mimicking a soft tissue sarcoma
*Corresponding author
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Primary cutaneous B-cell lymphomas (PCBCL) are a heterogeneous group of neoplasms with distinct biology and clinical course when compared to their nodal counterparts. They usually present as violaceous, erythematous plaques, and nonulcerated nodules, which are confined to skin at the time of presentation. We present an unusual case of primary cutaneous diffuse large B-cell lymphoma, clinically mimicking a sarcoma. This case highlights the uncommon aggressive behavior and ulcerated type of nodular lesions seen in PCBCL and also revisits the cytomorphological findings of the same.
Keywords
Cutaneous lymphoma
diffuse large B-cell lymphoma
fine needle aspiration cytology
non-Hodgkin's lymphoma
sarcoma
INTRODUCTION
Primary cutaneous lymphomas (PCLs) are usually confined to skin, present as patches, violaceous/erythematous plaques or nodules without ulceration and without evidence of extracutaneous disease at the time of diagnosis. However, cases with aggressive behavior are documented in literature.[1]
PCLs include cutaneous T-cell lymphomas and cutaneous B-cell lymphomas (CBCLs).[2] Primary CBCLs are less frequent and comprise only 20%–25% of all PCLs.[3] Primary cutaneous B-cell lymphomas are a heterogeneous group of lymphomas with distinct biology and clinical course when compared to their nodal counterparts. We discuss a case of an aggressive CBCL that clinically and radiologically mimicked soft tissue sarcoma.
CASE REPORT
A 40-year-old male presented with complaints of dull aching pain in the left leg for 4 months, limb edema and multiple progressively increasing swellings for 2 months. A month later, ulceration over the swellings developed and a rapidly increasing left inguinal region mass appeared. There was no history of fever, trauma, any other swelling in the body or systemic complaints. However, he gave a history of weight loss and decreased appetite. He was addicted to ganja and bhang. On examination, left lower limb was edematous and multiple firm nontender, subcutaneous nodular swellings ranging from 2 cm to 8 cm in largest diameter were noted. Three of these swellings, around knee joint were ulcerated [Figure 1a]. The ulcers had irregular margins and ragged base covered by necrotic slough. The left inguinal region swelling was subcutaneous, firm, nodular, and measured 8 cm in largest diameter. There was no other palpable peripheral lymphadenopathy. Per-abdomen and systemic examination did not reveal any abnormality. Hematological and biochemical parameters were within normal range. The provisional clinical diagnosis was soft tissue sarcoma.
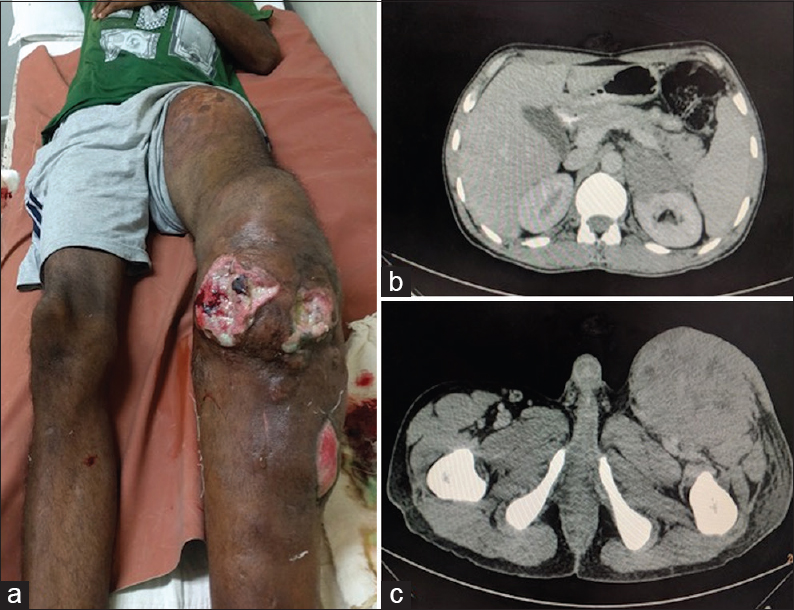
- (a) Left leg with multiple subcutaneous swellings and few ulcerated nodules. (b) Axial computed tomography: Homogeneously enhancing relatively hypodense left suprarenal mass with loss of fat plane in anterior pararenal space. (c) Axial computed tomography: Heterogeneously enhancing left inguinal lymph nodal mass
Fine-needle aspiration cytology (FNAC) from inguinal swelling and subcutaneous knee swelling was done; direct smears and cellblocks were prepared. Smears were air-dried and alcohol fixed and subsequently stained with Giemsa and Papanicolaou stain, respectively. Microscopy revealed dispersed population of predominantly intermediate to large-sized atypical lymphoid cells having high nucleus-cytoplasm ratio, irregular nuclear membrane, some with nuclear indentation and convolution, opened up chromatin and many showing conspicuous nucleoli. Occasional bi- and multi-nucleated cells were also noted. Background showed the presence of histiocytes and reactive lymphoid cells. Cytomorphological features were suggestive of Non-Hodgkins lymphoma, large cell type [Figure 2a–g]. Abdominal and pelvic contrast-enhanced computed tomography (CECT) scans revealed an ill-defined heterogeneously enhancing mass lesion, 6.8 cm × 5.7 cm × 5.2 cm in size in the left suprarenal gland, seen infiltrating into the upper pole of left kidney with loss of fat planes, likely metastasis [Figure 1b]. Another large heterogeneously enhancing mass lesion in the left inguinal region infiltrating into overlying skin and underlying muscles was noted, suggestive of neoplastic lymph node [Figure 1c]. CECT neck and thorax were normal.
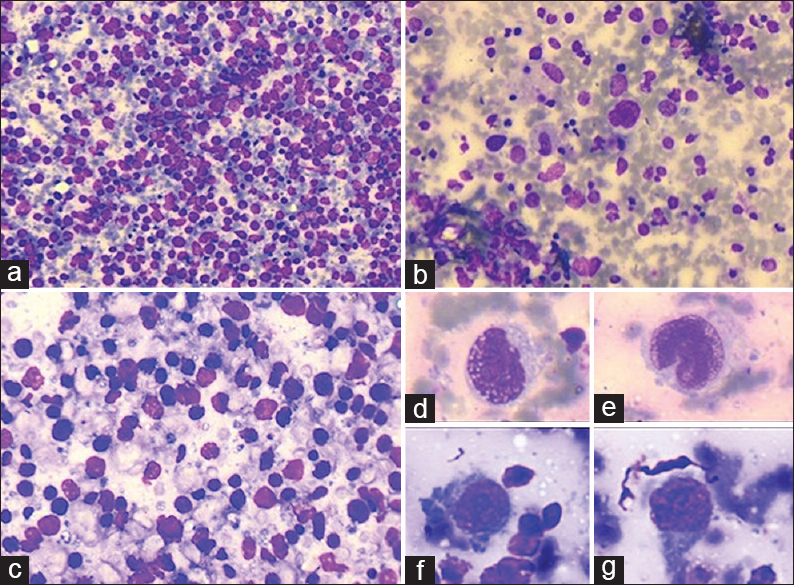
- (a) Cellular aspirates comprising dispersed population of intermediate to large lymphocytes (Giemsa, ×100). (b) Polymorphous background with scattered histiocytes (Giemsa, ×200). (c) Dispersed moderately pleomorphic atypical lymphoid cells (Giemsa, ×400). (d-g) Atypical large lymphoid cells with high-grade nuclear atypia; high nucleus-cytoplasm ratio, nuclear membrane irregularity, opened up chromatin, and prominent nucleoli (Giemsa, ×400)
Subsequently, skin biopsy revealed a pan-dermal infiltrate by sheets of large atypical lymphoid cells in the dermis with thinned out overlying epidermis. These lymphoid cells had scant cytoplasm, hyperchromatic nucleus with inconspicuous nucleoli. Mitosis was frequent and apoptosis evident [Figure 3a and b]. On immunohistochemistry the atypical lymphoid cells expressed leukocyte common antigen, CD20, and Bcl-6 [Figure 3c and d] while they were negative for CD3, CD5, CD10, CD23, CD30, cyclinD1, MUM-1, c-myc, Bcl-2, and EBER. The ki-67 index was approximately 80%. Histopathological diagnosis of primary cutaneous diffuse large B-cell lymphoma (DLBCL), other was made and c-myc/bcl-2 rearrangement studies were advised. Bone marrow aspiration was negative for tumor infiltration. The patient was referred to a higher oncology center for molecular analysis and an intensified chemotherapy regimen. However, he refused medical management and succumbed to his illness 2 months later.
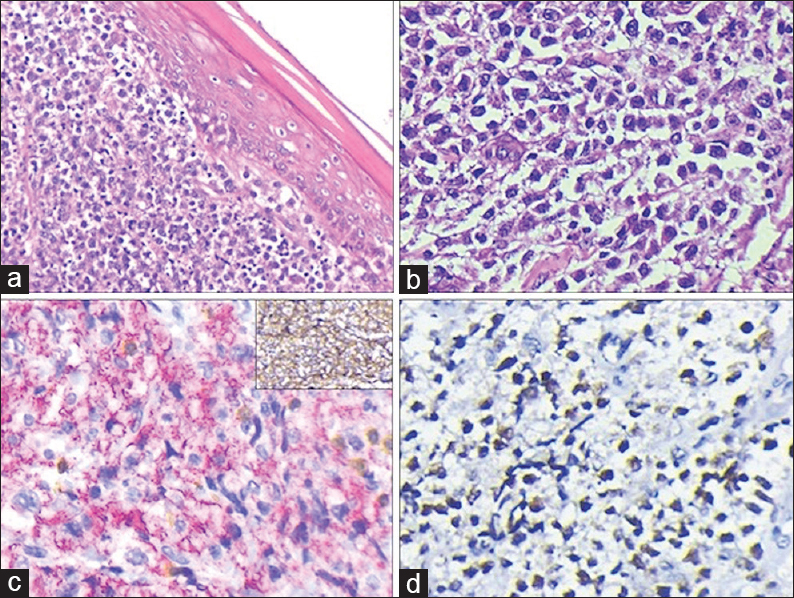
- (a) Tumor cells localized in the dermis (H and E, ×200). (b) Sheet and cords of atypical lymphoid cells displaying nuclear atypia (H and E, ×400). (c) CD20 positive tumor cells (Vulcan Fast Red Chromogen). Few scattered CD3 positive lymphocytes also seen. Inset: Leukocyte common antigen diffuse surface expression (DAB chromogen) (IHC, ×400). (d) Positive nuclear staining for Bcl-6 (IHC, ×400)
DISCUSSION
The 2008 World Health Organization adopted European Organization for Research Treatment of Cancer classification of cutaneous lymphomas that divides CBCLs as primary cutaneous marginal zone B-cell lymphoma, primary cutaneous follicle center lymphoma, primary cutaneous DLBCL, leg type (PCDLBCL, LT), and primary cutaneous DLBCL other (PCDLBCL, other).[45] PCDLBCL is uncommon aggressive lesions, which are difficult to characterize. The “other” category is broad and even more difficult to define histologically. This group includes rare morphological variants, such as anaplastic or plasmablastic subtypes, T-cell rich B-cell lymphoma, and some exceedingly rare DLBCL subtypes, such as intravascular large B-cell lymphoma and Epstein-Barr virus-associated large B-cell lymphoma of the elderly.[6] Also included are cases of CBCL with diffuse dermal infiltration of large cells not adaptable in the histopathological criteria for DLBCL, LT.
Primary cutaneous DLBCL is known to cause dissemination to uncommon extracutaneous sites.[7] In the present case of PCDLBCL, the patient had rapidly progressive clinical course and presented with unusual ulcerated type of lesions with extracutaneous dissemination, which was unilateral in spread with sparing of distant visceral sites such as bone marrow, lungs, liver, and brain. Clinically, it was mistaken for soft tissue sarcoma and an early FNAC resolved the dilemma. Whether the history of drug addiction can be attributable to rapid clinical course is uncertain.
Several therapeutic modalities are available for PCDLBCL; however, treatment should be selected taking into account the type of lymphoma, its stage, and the risk adapted score as per the International Prognostic Index.[8] Aggressive PCDLBCL is treated with multidrug therapy of CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) or CHOP-like regimens.[9]
We reiterate that cutaneous large B-cell lymphomas are at times difficult to characterize morphologically with many fitting in the “other” category. They have a wide spectrum of clinical presentation and ulcerated nodules with unilateral limb edema is one of them as seen in the current case.
Declaration of patient consent
The authors certify that they have obtained appropriate patient consent form. In the form, the patient has given his consent for his images and other clinical information to be reported in the journal. The patients understands that name and initials will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
COMPETING INTERESTS STATEMENT BY ALL AUTHORS
All authors declare that there are no competing interests.
AUTHORSHIP STATEMENT BY ALL AUTHORS
PG - was involved in conceptualizing, writing and editing of this manuscript; PA - was involved in writing, literature search and editing; AA - was involved in conceptualization and editing; CK - provided patient information and helped in editing of manuscript.
ETHICS STATEMENT BY ALL AUTHORS
This report does not require approval from Institutional Review Board.
LIST OF ABBREVIATIONS (In alphabetic Order)
DLBCL: Diffuse Large B-cell Lymphoma
NHL: Non-Hodgkin's Lymphoma
PCBCL: Primary Cutaneous B-cell Lymphoma
PCL: Primary Cutaneous Lymphoma.
EDITORIAL/PEER REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double-blind model (authors are blinded for reviewers and vice versa) through automatic online system.
REFERENCES
- Primary cutaneous diffuse large B-cell lymphoma: A clinicopathologic study of 15 cases. Am J Clin Pathol. 2002;117:574-80.
- [Google Scholar]
- Primary cutaneous large B-cell lymphoma of atypical presentation: Case report. An Bras Dermatol. 2011;86:549-51.
- [Google Scholar]
- Aliterature revision in primary cutaneous B-cell lymphoma. Indian J Dermatol. 2017;62:146-57.
- [Google Scholar]
- Lymphoma presenting as a musculoskeletal soft tissue mass: MRI findings in 24 cases. Eur Radiol. 2008;18:2628-34.
- [Google Scholar]
- Cutaneous primary B-cell lymphomas: From diagnosis to treatment. An Bras Dermatol. 2015;90:687-706.
- [Google Scholar]








