Translate this page into:
The International Academy of Cytology standardized reporting of breast fine-needle aspiration biopsy cytology: A 2 year’s retrospective study with application of categories and their assessment for risk of malignancy

-
Received: ,
Accepted: ,
How to cite this article: Nigam JS, Kumar T, Bharti S, Surabhi, Sinha R, Bhadani PP. The International Academy of Cytology standardized reporting of breast fine-needle aspiration biopsy cytology: A 2 year’s retrospective study with application of categories and their assessment for risk of malignancy. CytoJournal 2021;18:27.
Abstract
Objectives:
Breast cancer is the most common cancer in women worldwide. The fine-needle aspiration biopsy (FNAB) may be used as the first-line pathological investigation for evaluation and early diagnosis of the breast lesion. The FNAB helps to differentiate malignant from benign lesions. In the present study, we categorized the breast FNAB cases according to the International Academy of Cytology Yokohama System (IACYS) for reporting breast FNAB cytology and to assess the risk of malignancy (ROM) for each category.
Material and Methods:
A retrospective data of breast lesions were retrieved from the archives of pathology department between January 2018 and December 2019. The study got approval from the Institutional Ethics Committee. Only 123 cases with cytology and histopathological correlation were included in this study. The cytological category was given according to IACYS for reporting breast FNAB cytology.
Results:
The FNAB results were include as insufficient material 3.25% (4/123), benign 46.34% (57/123), atypical 12.2% (15/123), suspicious for malignancy (SM) 4.88% (6/123), and malignant 33.33% (41/123). The ROM was 50%, 7.27%, 40.0%, 83.33%, and 97.5% for NS, benign, atypical, SM, and malignant, respectively.
Conclusion:
FNAB is an important tool in the diagnosis and management of breast lesions, especially in financial constrained developing countries like India with limited resources, where practice of core needle biopsy is limited. The 5-tier IACYS for reporting breast FNAB improves the reproducibility of cytology reports across the world and helps in triaging the breast lesion patients.
Keywords
Breast
Cytology
Cancer
Cytopathology
India
Women
Benign neoplasms
Tumors
INTRODUCTION
Breast cancer (BC) is the most common cancer in women worldwide.[1] Due to increased life expectancy, increased urbanization, and adoption of western lifestyles, the incidence of BC is increasing in the developing world.[1] In India, it accounts for 14% of all cancers in female with peak ages of 50–64 years and shows a rise in the rate of incidence in the early 30s.[2]
In developing countries like India, majority of BC is diagnosed in late stages, and therefore, early detection is the keystone to improve BC management and outcome.[1] There are various investigations modalities available for the diagnosis of breast masses.[3] However, the triple test is the forerunner investigation used in both screening and in symptomatic patients.[3,4] Fine-needle aspiration biopsy (FNAB) cytology is a minimally invasive and better tolerable diagnostic technique for initial evaluation of breast lesion as it provides rapid and accurate diagnosis at low cost.[4] The FNAB and core needle biopsy (CNB) are the complimentary tests, both have their pros and cons.[4,5] FNAB may help to differentiate malignant from benign lesions, however, differentiation is not achieved in all the cases due to a significant cytological features overlap.[3] In 1996, the National Cancer Institute proposed five diagnostic categories, unsatisfactory (C1), benign (C2), atypical, probably benign (C3), suspicious, favor malignancy (C4), and malignant (C5) to bring an uniform reporting nomenclature.[3] After two decades, the International Academy of Cytology (IAC) Breast Group was brought together in 2016 at the Yokohama International Congress of Cytology with the aim to develop an internationally acceptable, standardized, and uniform reporting system, henceforth, the guidelines were proposed for the use of FNAB in diagnosing breast lesions.[4-6] This system may help to improve the reporting quality, interpretation, and clarify the message to clinicians and also improves the reproducibility of breast cytology reports across the world for better patient management.[5,6] The IAC Yokohama System (IACYS) for reporting breast FNAB cytology has 5-tire system.[4-6] The following categories are proposed by IACYS for reporting breast FNAB cytology: Category (1) insufficient; category (2) benign; category (3) atypical; category (4) suspicious of malignancy; and category (5) malignant.[4-6] In the present study, breast cytology cases reported in our institute were categorized according to the IACYS for reporting breast FNAB cytology. The main objectives of the study were to categorize the breast FNAB samples according to this newly proposed system and to assess the risk of malignancy (ROM) for each category.
MATERIAL AND METHODS
A retrospective data of breast lesions were retrieved from the archive of department of pathology between January 2018 and December 2019. Only 123 cases with both histological and cytological diagnosis were included in this study. This study was approved by the Institutional Ethics Committee vide letter no. AIIMS/Pat/IEC/2020/444. The patients of all age groups were included in the study. Fine-needle aspiration (FNA) was performed under aseptic precaution using a 10 ml disposable syringe and 23/24-gauge needle. The air-dried smears were stained with Giemsa stain while those fixed in 95% alcohol were stained by Papanicolaou’s stain. The cases were evaluated according to the newly proposed “IACYS for reporting breast FNAB cytology 2016.”
For histopathology examination, 10% neutral-buffered formalin-fixed, surgically resected specimen or biopsy tissues were processed to make paraffin-embedded tissue blocks. Sections of 3–4 µ were cut from block and stained with hematoxylin and eosin stain. The results of cytology and histopathology were compared considering the histopathology as the gold standard for final diagnosis. The statistical analysis was done for sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and diagnostic accuracy. The ROM was calculated by dividing the total number of cases turned out to be malignant on histopathology in each category by total number of cases in each category on cytology.
RESULTS
One hundred and twenty-three cases of cytologically diagnosed breast lesions were included in this study and correlated with corresponding paraffin-embedded sections. Size of the breast lesions was varied from <1 cm to 19 cm in greatest dimension. The most common age group were 31–40 years. Only one male patient was present in this study. The most common presentation was that of a breast lump, with slightly left side predominance (50.4%, 62/123) [Figure 1]. Upper outer quadrant was the most frequently involved site (29.27%, 36/123) followed by central quadrant (21.95%, 27/123). Twenty-two cases (17.89%) involved more than 1 quadrant [Figure 2]. The FNA cytology (FNAC) results showed that 3.25% (4/123) of cases were insufficient/ inadequate, 46.34% (57/123) benign, 12.2% (15/123) atypical, 4.88% (6/123) suspicious for malignancy, and 33.33% (41/123) of cases were malignant.
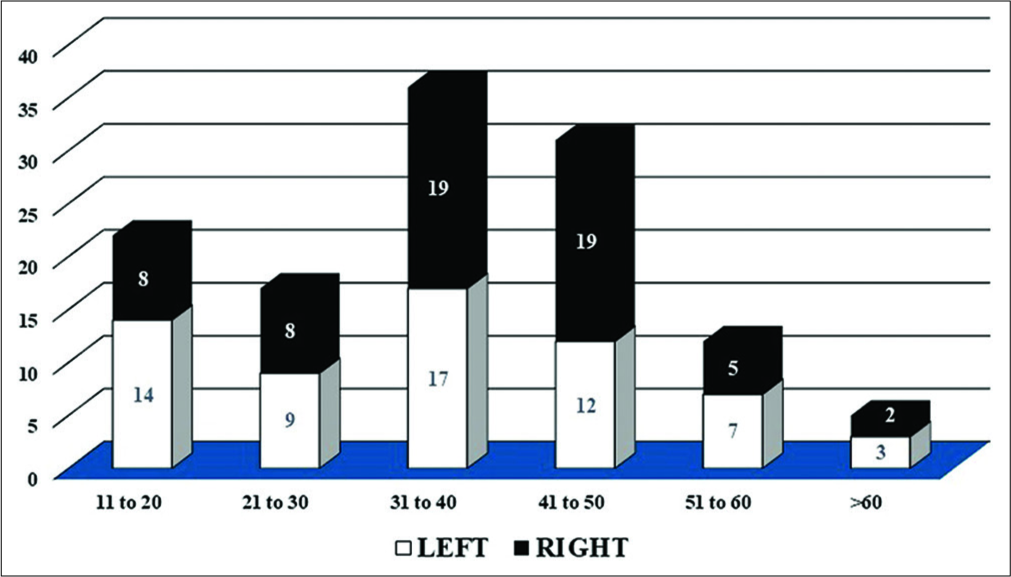
- Age- and side-wise distribution of breast lesions.

- Quadrant-wise distribution of breast lesions.
In category 1 [Figure 3] (insufficient/inadequate), two cases with lesion <1 cm were also inadequate on biopsy. One lesion turned out to be malignant histopathology and it was >5 cm in size but sufficient material was not aspirated even on repeated attempts. The ROM of 50% (1/2) was considered.

- (a) Non-interpretable aspirate with occasional small cluster of epithelial cell (Giemsa ×40), (b) Poor aspirate with predominant blood (Giemsa ×40).
Out of 57 cases of category 2 [Figure 4], two cases had non-representative material, three cases showed malignancy, and one case turned out to be borderline phyllodes on histological examination. The ROM was 7.27% (4/55, three malignant and one borderline phyllodes).

- (a) Predominant necrotic aspirate (Giemsa ×100), (b) Sparsely cellular smear with an occasional macrophage in thin proteinaceous background (Giemsa ×400), (c) Cohesive clusters and flat monolayered sheets of ductal epithelial cells with myoepithelial cells along with bare bipolar nuclei and myxoid stromal fragment (Giemsa ×40), (d) Numerous spindle-shaped stromal cells along with few cluster of ductal epithelial cells admixed with myoepithelial cells (Giemsa ×100), (e) Granulomatous mastitis: Granuloma in necrotic background and few ductal epithelial cells (Papanicolaou ×400), (f) Filarial worm in inflammatory background with few ductal epithelial cells (Papanicolaou ×400).
In category 3 [Figure 5], one case of atypical proliferation on cytology showed benign papillary lesion and six cases were turned out to be malignant on histology. The ROM was 40.0% (6/15).

- (a) Scanty cellular smear shows an occasional papillary fragment with central fibrovascular core (Papanicolaou ×100, Inset: Papanicolaou ×400), (b) Sparsely cellular smear shows dispersed and few clusters of ductal epithelial cells having mild nuclear atypia in the mucinous background (Giemsa ×100, Inset: Giemsa ×400), (c) Ductal epithelial cells showing mild-to-moderate atypia with few macrophages in the proteinaceous background (Papanicolaou ×400), (d) Cluster of benign ductal epithelial cells along with small cluster of cells showing mild-to-moderate nuclear atypia (Papanicolaou ×400).
The ROM in category 4 [Figure 6] was 83.33% (5/6, four malignant and one DCIS). Only one case showed benign histology.

- (a) Cluster of ductal epithelial cells along with cluster of cells showing moderate-to-severe nuclear atypia (Giemsa ×400), (b) Cellular smear with moderate nuclear atypia (Giemsa ×400), (c) Scant cellular aspirate with severe nuclear atypia (Papanicolaou ×100, Inset: Papanicolaou ×400), (d) Scant cellular aspirate show oval- to spindle-shaped cells severe nuclear atypia (Giemsa ×400).
In category 5 [Figure 7], all cases were malignant on both histology and cytology except two cases. One case showed inadequate biopsy tissue and the other case with lesion of <1 cm turned out benign on histopathology. The ROM was 97.5% [Table 1].
| Cytological category (no. of cases) |
Histology (no. of cases) | |||
|---|---|---|---|---|
| Benign | Malignant | Borderline | % ROM | |
| Insufficient/non-diagnostic | 1 | 1 | 0 | 50 |
| Benign | 51 | 3 | 1 | 7.27 |
| Atypical | 9 | 6 | 0 | 40 |
| Suspicious | 1 | 4 | 1 | 83.33 |
| Malignant | 1 | 39 | 0 | 97.5 |
ROM: Risk of malignancy

- (a and b) Cellular smears with discohesive cells showing moderate-to-severe nuclear atypia (Giemsa ×100, Papanicolaou ×400), Papillary fragment with moderate nuclear atypia (Papanicolaou ×100), (c) Discohesive cells showing moderate-to-severe nuclear atypia having vesicular to salt and pepper chromatin (Papanicolaou ×400).
The sensitivity, specificity, PPV, NPV, and diagnostic accuracy were evaluated by grouping the insufficient, benign, atypical, suspicious, and malignant into three different groups and comparing with malignant histology with or without borderline/in situ carcinomas. Group A included only malignant cytology cases, Group B included suspicious and malignant cytology cases while Group C included atypical, suspicious, and malignant cytology cases. These groups were compared with Class 1 (malignant histology only) and Class 2 (malignant with borderline/in situ component). We observed that specificity and PPV were highest in Group A in both classes. While, sensitivity and NPV were highest in Group C in both classes [Table 2].
| Malignant histology only | |||||
|---|---|---|---|---|---|
| Sensitivity | Specificity | PPV | NPV | Accuracy | |
| Group A | 73.58 | 98.46 | 97.5 | 82.05 | 87.29 |
| Group B | 81.13 | 95.38 | 93.48 | 86.11 | 88.98 |
| Group C | 92.45 | 81.54 | 80.33 | 92.98 | 86.44 |
|
Malignant histology (malignant + borderline + in situ carcinoma) |
|||||
| Sensitivity | Specificity | PPV | NPV | Accuracy | |
| Group A | 70.91 | 98.41 | 97.5 | 79.49 | 85.59 |
| Group B | 80.00 | 96.83 | 95.65 | 84.72 | 88.98 |
| Group C | 90.91 | 82.54 | 81.97 | 91.23 | 86.44 |
PPV: Positive predictive value, NPV: Negative predictive value
DISCUSSION
The breast lesions comprise different benign and malignant lesions with the gray-zone lesions between them due to overlap in their cytological features.[3,7] The benign lesions are more common than malignant lesions.[7] The triple test encompasses of clinical examination, mammography/ ultrasonography (USG), and FNAB/CNB which help to make an accurate diagnosis in approximately 99% of malignant cases.[7,8] FNAB is the important tool to play a major role in the diagnosis and management of breast lesions, especially in financial constrained countries like India with limited resources, where practice of CNB is limited.[7,8] The FNAB is a cost-effective technique which helps to obtain the sample from various sites of lesion in a single visit without extra financial burden.[7,8] The rapid onsite evaluation helps to reduce the inadequacy rate for material and also triaging the cases for other ancillary diagnostic workup.[4,7-9] Similar to the other organs’ cytopathology reporting systems, IACYS for reporting breast FNAC will helps improving the reproducibility of reports across the world and also facilitate the communication to the clinician for their optimal patient management.[5-9]
In the present study, we retrospectively adopted the newly proposed IACYS for reporting breast FNAB cytology and categorized into category 1–category 5. The category 1 is defined as “the smears are too sparsely cellular or too poorly smeared or fixed to allow a cytomorphological diagnosis.” If FNAB material does not explain the imaging or clinical findings, the report should make a note that “the material may not represent the lesion.”[4-6] Category 1 constitutes from 0.69% to 35.43% of breast FNAB and ROM is ranged from 0% to 83.3%.[3,4,7-10] In the present study, category 1 constituted 3.25% of all cases with ROM of was 50%. The various factors which affects the rate of inadequacy are nature of the lesion, available technology, experience, and skill of the performer.[3,6] However, to lower the inadequacy rate, some studies recommend that both FNAB performer and interpreter should ideally be same to increase the diagnostic yield.[3] Category 2 is characterized by predominantly large cohesive, three-dimensional tissue fragments, and flat monolayered sheets in bimodal pattern with many bare bipolar nuclei in the background.[5-6] These cytological features may or may not be definitive of a specific benign lesions, however, diagnosis with negative clinical and radiological findings is considered as adequate diagnostic workup.[5-6] The cases of category 2 vary from 4.65% to 84.17% with ROM are ranged from 0% to 33.3%.[3,4,6-10] We observed that category 2 constituted 46.34% cases with ROM of 7.27%. The category 3 is characterized by the presence of predominantly benign cytological features with some features that are uncommon in benign lesions which may be seen in malignant lesions such as high cellularity, increased dispersed cells, nuclear pleomorphism, necrosis, mucin, complex micropapillary, and cribriform architecture.[5,6] Around 2.1–20.21% of all breast FNABs come under category 3 with ROM varying from 0% to 70.5%.[3,4,6-10] In the present study, ROM for category 3 was 42.86% constituting 12.2% of all cases. Suspicious of malignancy (category 4) shows the presence of insufficient cytomorphological malignant features, either in number or quality, to make a definitive diagnosis of malignancy.[5,6] The cytomorphological features are highly cellular smears with epithelial tissue fragment showing cribriform, micropapillary or papillary architecture, increased dispersed cells with mild-to-moderate nuclear atypia, reduced or absent myoepithelial cells as well bare bipolar nuclei in the background.[5,6] Smears with low cellularity showing high-grade nuclear atypia, necrosis, and calcifications may also be included in category 4.[5,6]
The category 4 represents 1.53–25.89% and ROM varies from 26.7% to 100%.[3,4,6-10] The present study revealed 4.88% of cases of category 4 with 83.33% ROM. The rate of category 5 varies from 6.19% to 70.93% with ROM varies from 91.2% to 100%.[3,4,6-10] The major cytomorphological features included are high cellularity, prominent dispersal of single cells, crowded tissue fragments with overlapping nuclei, nuclear enlargement, anisonucleosis, pleomorphism of the nuclear margin, size and chromatin, hyperchromasia, and prominent nucleoli.[5,6] In the present study, category 5 represents 33.33% of cases and has 97.5% of ROM. The previous studies of breast FNAB cytology show the sensitivity ranging from 76% to 99.3%, specificity from 25% to 100%, PPV from 74% to 100%, NPV from 25% to 99.1%, and diagnostic accuracy 72–99%.[3,4,7-10] We divided the cases in three groups, that is, A, B, and C based on cytology and two classes based on histology findings [Table 2]. Our results were in concordance with Montezuma et al.[7] with highest sensitivity and NPV in Group C and highest specificity and PPV in Group A in both the classes. The main limitations of the present study are its small sample size and high ROM in insufficient, benign, and atypical category as compared to recommendations given by Field et al.[6] This could be attributed to the sampling error which may be overcome by USG-guided procedure as recommended by IACYS reporting for breast FNAB.
CONCLUSION
FNAB is the important tool to play major role in diagnosis and management of breast lesions especially in financial constraints, developing countries like India. The 5-tier IACYS for reporting breast FNAB cytology will help improving the reproducibility of reports and facilitate the communication to the clinician for better management.
Funding sources
No funding source.
COMPETING INTEREST STATEMENT BY ALL AUTHORS
The authors have no conflicts of interest to declare.
AUTHORS’ CONTRIBUTIONS
Drafting of manuscript: Tarun Kumar, Jitendra Singh Nigam, Shreekant Bharti, and Surabhi; literature search: Tarun Kumar, Jitendra Singh Nigam, Shreekant Bharti, and Surabhi; study conception/design: Tarun Kumar, Jitendra Singh Nigam, Ruchi Sinha, and Punam Prasad Bhadani; data acquisition: Tarun Kumar, Jitendra Singh Nigam, Surabhi, and Ruchi Sinha; data analysis and interpretation: Tarun Kumar, Jitendra Singh Nigam, Shreekant Bharti, Surabhi, Ruchi Sinha, and Punam Prasad Bhadani; manuscript preparation: Tarun Kumar and Jitendra Singh Nigam; manuscript editing: Tarun Kumar, Jitendra Singh Nigam, and Shreekant Bharti; and manuscript review: Tarun Kumar, Jitendra Singh Nigam, and Punam Prasad Bhadani.
ETHICS STATEMENT BY ALL AUTHORS
This study was approved by the Institutional Ethics Committee vide letter no. Ref.No.AIIMS/Pat/IEC/2020/444.
LIST OF ABBREVIATIONS (IN ALPHABETIC ORDER)
BC: Breast Cancer
CNB: Core needle biopsy
FNA: Fine-needle aspiration
FNAB: Fine-needle aspiration biopsy
FNAC: Fine-needle aspiration cytology
IAC: International Academy of Cytology
IACYS: International Academy of Cytology Yokohama System
NPV: Negative predictive value
PPV: Positive predictive value
ROM: Risk of malignancy
SM: Suspicious for Malignancy
EDITORIAL/PEER-REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double-blind model (authors are blinded for reviewers and vice versa) through automatic online system.
References
- Available from: https://www.who.int/cancer/detection/breastcancer/en [Last accessed on 2020 Jun 15]
- Available from: http://www.cancerindia.org.in/cancer-statistics [Last accessed on 2020 Jun 15]
- Application of national cancer institute recommended terminology in breast cytology. J Cancer Res Ther. 2017;13:91-6.
- [CrossRef] [PubMed] [Google Scholar]
- The international academy of cytology Yokohama system for reporting breast fine needle aspiration biopsy cytopathology: A single institutional retrospective study of the application of the system categories and the impact of rapid onsite evaluation. Acta Cytol. 2019;63:280-91.
- [CrossRef] [PubMed] [Google Scholar]
- IAC standardized reporting of breast fine-needle aspiration biopsy cytology. Acta Cytol. 2017;61:3-6.
- [CrossRef] [PubMed] [Google Scholar]
- The international academy of cytology Yokohama system for reporting breast fine-needle aspiration biopsy cytopathology. Acta Cytol. 2019;63:257-73.
- [CrossRef] [PubMed] [Google Scholar]
- Breast fine needle aspiration biopsy cytology using the newly proposed IAC Yokohama system for reporting breast cytopathology: The experience of a single institution. Acta Cytol. 2019;63:274-9.
- [CrossRef] [PubMed] [Google Scholar]
- FNAC of breast lesions with special reference to IAC standardized reporting and comparative study of cytohistological grading of breast carcinoma. J Cytol. 2020;37:34-9.
- [CrossRef] [PubMed] [Google Scholar]
- International academy of cytology Yokohama system for reporting breast fine-needle aspiration biopsy cytopathology: A review of predictive values and risks of malignancy. Acta Cytol. 2019;63:292-301.
- [CrossRef] [PubMed] [Google Scholar]
- IAC standardized reporting of breast fine-needle aspiration cytology, Yokohama 2016: A critical appraisal over a 2 year period. Breast Dis. 2019;38:109-15.
- [CrossRef] [PubMed] [Google Scholar]









