Translate this page into:
The effectiveness of acetic acid wash protocol and the interpretation patterns of blood contaminated cervical cytology ThinPrep® specimens
*Corresponding author
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background:
ThinPrep® (TP) cervical cytology, as a liquid-based method, has many benefits but also a relatively high unsatisfactory rate due to debris/lubricant contamination and the presence of blood. These contaminants clog the TP filter and prevent the deposition of adequate diagnostic cells on the slide. An acetic acid wash (AAW) protocol is often used to lyse red blood cells, before preparing the TP slides.
Design:
From 23,291 TP cervical cytology specimens over a 4-month period, 2739 underwent AAW protocol due to initial unsatisfactory smear (UNS) with scant cellularity due to blood or being grossly bloody. Randomly selected 2739 cervical cytology specimens which did not undergo AAW from the same time period formed the control (non-AAW) group. Cytopathologic interpretations of AAW and non-AAW groups were compared using the Chi-square test.
Results:
About 94.2% of the 2739 cases which underwent AAW were subsequently satisfactory for evaluation with interpretations of atypical squamous cells of undetermined significance (ASCUS) 4.9% (135), low-grade squamous intraepithelial lesions (LSIL) 3.7% (102), and high-grade squamous intraepithelial lesions (HSIL) 1% (28). From the 2739 control cases, 96.3% were satisfactory with ASCUS 5.5% (151), LSIL 5.1% (139), and HSIL 0.7% (19). The prevalence of ASCUS interpretations was similar (P = 0.33). Although there were 32% more HSIL interpretations in the AAW group (28 in AAW vs. 19 in non-AAW), the difference was statistically insignificant (P = 0.18). AAW category; however, had significantly fewer LSIL interpretations (P = 0.02). The percentage of UNS cases remained higher in the AAW group with statistical significance (P < 0.01).
Conclusions:
While AAW had a significantly higher percent of UNS interpretations, the protocol was effective in rescuing 94.2% of specimens which otherwise may have been reported unsatisfactory. This improved patient care by avoiding a repeat test. The prevalence of ASCUS and HSIL interpretations between AAW and non-AAW groups were comparable. Though not statistically significant, HSIL interpretations were relatively higher in the AAW group. LSIL interpretations showed lower prevalence in AAW group.
Keywords
Acetic acid wash
cervical cytology
ThinPrep®
INTRODUCTION
The use of liquid-based cytology has many advantages over the conventional smears including better cytomorphology with lack of air-drying artifacts allowing a higher detection rate of squamous epithelial abnormalities. This is especially true for high-grade squamous intraepithelial lesions (HSIL), reaching up to a 233% increase in HSIL detection in one study.[1] ThinPrep® (TP) is one of the liquid-based preparation for evaluation of cervical specimens as Papanicolaou (Pap) test. An earlier large study in 1999 by Dνaz-Rosario and Kabawat showed a 102% increase in HSIL detection with the TP Pap Test versus the conventional Pap smear.[2] Both studies also found increases in low-grade squamous intraepithelial lesions (LSIL) detection as well.[12]
Another important advantage of the TP liquid-based preparation is the reduction in the unsatisfactory rate in comparison to conventional smear. Lee et al., showed a significant increase in both the satisfactory specimens as well as a reduction in the number of “satisfactory but limited by” specimens using the TP method.[3]
A TP Pap Test slide is clearer, easier to read, and minimizes obscuring blood, mucus, and nondiagnostic debris, compared to a conventional smear enabling increased accuracy for both manual assessment and computerized assessment of the cells. However, since the adoption of the liquid-based cytology in many laboratories across the US, there are still a measurable number of unsatisfactory smears (UNS).[4] One of the well-known and documented causes of unsatisfactory material is blood contaminated cervical cytology specimens which not only affects interpretation, but may also hide significant epithelial abnormalities.[45]
Many cytology laboratories in the US have implemented an acetic acid wash (AAW) method of reprocessing unsatisfactory PreservCyt samples and have shown that this method reduces the unsatisfactory rate and enhances the ability to detect cellular abnormalities in these specimens. The studies by Bentz et al.[6] and Islam et al.[7] showed AAW could reduce unsatisfactory TP cytology results by approximately 62% and could detect epithelial abnormalities in reprocessed TP specimens. The most common epithelial abnormality identified in these specimens was atypical squamous cells of undetermined significance (ASCUS).[67] Figure 1 shows photomicrographs of a case, which was unsatisfactory due to scant cellularity and obscuring by blood and became satisfactory after AAW.
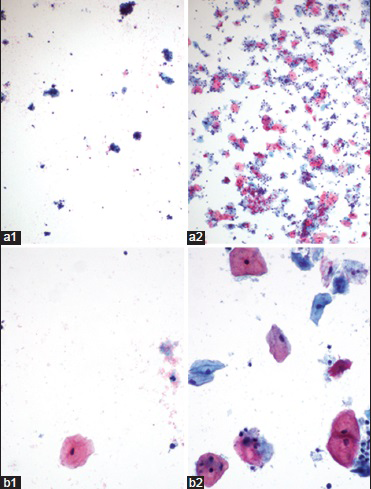
- Images of pre- (1) and post (2)-acid wash ThinPrep® cervical cytology specimens (a, ×4; b, ×40)
Importantly, a study from our institution by Feng and Husain[8] showed that processing of bloody specimens with AAW did not appear to significantly affect high-risk human papillomavirus (HPV) DNA testing in specimens with an ASCUS interpretation and no statistically significant difference were also noted with regards to age, percentage HPV DNA positivity, or viral load between pretreated and untreated cases. Feng and Husain also showed that the unsatisfactory rate was reduced from 8% to 3%.[8] Comparable reduction rates were reported by other laboratories.[67]
In the current study, we evaluated the adequacy and final cytopathologic interpretation patterns among the specimens undergoing AAW protocol and compared the results with randomly matched non-AAW specimens reported during the same time period.
MATERIALS AND METHODS
Out of 23,291 TP cervical cytology specimens over a 4 months period, 2739 underwent the AAW protocol because they were initially UNS with scant cellularity due to blood or were grossly bloody. We then randomly selected a matched set of 2739 cases which did not undergo AAW from the same 4-month time period. The matched group was selected both temporally and by number of cases. This would allow for allow better analysis of the differences in the rates of abnormal interpretations and adequacy status.
We reviewed diagnostic reports of the cases for adequacy and final interpretation as UNS, ASCUS, LSIL, or HSIL. The data were statistically analyzed in order to compare the AAW group and the control non-AAW group. The Chi-square test was used to determine statistical significance.
The details of the AAW protocol used in our laboratory are as follows. The specimen is poured into a 50 ml centrifuge tube and is centrifuged for 10 min at 1500 rpm. The supernatant is carefully decanted and discarded. Thirty milliliter of CytoLyt/glacial acetic solution is added to the tube and the tube is vortexed for 10 s. The specimen is then centrifuged at 1500 rpm for another 10 min. The supernatant is again decanted and discarded, and the cell button is resuspended in 20 ml PreservCyt medium and then processed as a usual TP specimen.
RESULTS
Of the 23,291 TP cervical cytology specimens 5478 were included in the study. These included 2739 cases that underwent AAW protocol since they were UNS with scant cellularity due to blood or were grossly bloody at initial examination (AAW set). In addition, a control group of 2739 matched cases which did not undergo AAW protocol (control group) were also included in the study. The matching of the study case sets was done both temporally and by the number of cases in order to allow better analysis of the differences in the rates of abnormal interpretations and adequacy status.
Of the 2739 “AAW” cases that underwent AAW protocol, 2580 (94.2%) were subsequently satisfactory for evaluation. The cytopathologic interpretations of these cases were: 135 (4.9%) ASCUS, 102 (3.7%) LSIL, and 28 (1%) HSIL. In the matched “control” group of 2739 cases which did not undergo AAW protocol, 96.3% were satisfactory for evaluation with 151 (5.5%) ASCUS, 139 (5.1%) LSIL, and 19 (0.7%) HSIL [Table 1]. The difference in the cytopathologic interpretation patterns between the two groups, study set and control groups, was statistically insignificant (P = 0.33) for ASCUS. Figure 2 shows photomicrographs of cases in each interpretive category.
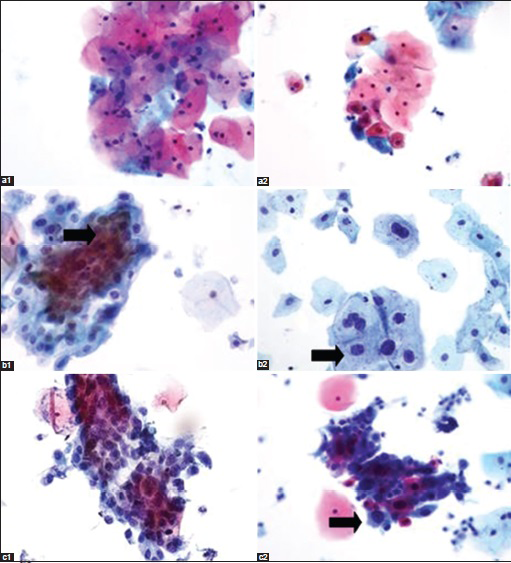
- Images of post acid wash (1) and nonacid wash (2) ThinPrep® cervical cytology specimens with abnormalities (a, atypical squamous cells of undetermined significance × 40; b, low-grade squamous intraepithelial lesions × 40; c, high-grade squamous intraepithelial lesions ×40)
Although there were 32% more HSIL cases in the AAW group (28 in AAW vs. 19 in the non-AAW control group), the difference was statistically not significant (P = 0.18). The AAW group of cases, however, had relatively fewer cases with LSIL interpretations as compared to the control group with statistically significant difference (P = 0.02).
Further despite performing the AAW protocol, the percentage of UNS cases remained relatively higher in the AAW group (5.8% in AAW vs. 3.7% in non-AAW control group). This was statistically significant (P < 0.01).
DISCUSSION
The AAW protocol is clearly of great clinical benefit. However, the unsatisfactory rate remained higher in the AAW group compared to the control group. The satisfactory rate of 94.2% in the AAW group was significantly higher than the specimens with blood contamination at the initial examination. This improvement represented a great cost reduction in clinical care by preventing a return clinic visit and repeat Pap test for many women at our institution. This benefit has also been reported by many other publications.[5679101112]
This is the first study, to our knowledge, in which a set of cases undergoing a pretreatment with acetic acid was compared to a matched set of cases which did not undergo the AAW protocol in order to compare interpretation patterns. The matching of the case sets both temporally and by the number of cases, allowed us to better analyze the differences in the rates of abnormal interpretations and adequacy status.
The rate of ASCUS interpretations for both the groups were comparable (5.5% for AAW vs. 4.9% control cases, P = 0.33). Previous studies demonstrate the pretreatment with acetic acid (AAW) does not hinder the use of the TP residual specimen for HPV DNA testing. This is a critical ancillary test in the clinical triage of gynecology patients to colposcopy with the diagnosis of ASCUS.[813]
There were 32% more HSIL interpretations in the AAW group (28, 1%) as compared to the control non-OA group (19, 0.7%). However, this difference was statistically insignificant (P = 0.18). The statistical insignificance may be due to the low number of HSIL cases in comparison to the rather large denominator of thousands of samples.
A few other studies have shown higher rates of high-grade lesions, as well as glandular lesions, and cancer in bloody Pap smear samples compared to without blood contamination.[561214] This finding makes the use of AAW protocol even more important, as missing a high-grade lesion could be of great detriment to the patient.
The prevalence of LSIL interpretation was lower in the AAW group (3.7%) as compared to the control non-AW group (5.1%) with statistically significant difference (P = 0.02). Although the explanation for this finding was not obvious, one potential reason may be that LSIL clinically do not produce bloody TP samples as often as other cervical lesions. Another possibility is that somehow the AAW protocol itself is directly decreasing the rate of interpretation of LSIL, possibly by altering the morphology of the dysplastic cells. Cohen et al., reported that AAW increased the false-positive rate of atypical endocervical glandular lesions by demonstrating that on follow-up the AAW treated cases had significantly fewer lesions on biopsy than the control group.[15] They concluded that the AAW protocol affected the morphology of the endocervical cells in the sample, thereby leading to false-positive interpretations.[15] This may suggests that AAW protocol could also potentially alter the morphology of squamous epithelial cells.
CONCLUSION
Our findings demonstrate that the pretreatment with acetic acid is of great benefit in reducing the unsatisfactory rate of bloody cervical cytology specimens. The rate of interpretation of ASCUS was the same between AAW and control groups. The rate of HSIL was relatively higher in the AAW group, and the rate of LSIL was significantly lower. Further studies may be needed to help elucidate the finding of a relatively decreased number of LSIL in AAW group.
COMPETING INTERESTS
All authors declare that they have no competing interests.
AUTHORSHIP STATEMENT BY ALL AUTHORS
All authors of this article declare that we qualify for authorship as defined by ICMJE http://www.icmje.org/#author.
Each author has participated sufficiently in the work and takes public responsibility for appropriate portions of the content of this article.
All authors read and approved the final manuscript.
Each author acknowledges that this final version was read and approved.
ETHICS STATEMENT BY ALL AUTHORS
This study was conducted with approval from Institutional Review Board (IRB) of all the institutions associated with this study as applicable.
LIST OF ABBREVIATIONS (In alphabetic order)
AAW - An Acetic Acid Wash
ASCUS - Atypical Squamous Cells of Undetermined Significance
HPV - Human Papillomavirus
HSIL - High-Grade Squamous Intraepithelial Lesions
IRB - Institutional Review Board
LSIL - Low-Grade Squamous Intraepithelial Lesions
Pap - Papanicolaou
TP - ThinPrep
UNS - Unsatisfactory Smear
EDITORIAL/PEER-REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double blind model (authors are blinded for reviewers and vice versa) through automatic online system.
Available FREE in open access from: http://www.cytojournal.com/text.asp?2015/12/1/23/168060
REFERENCES
- Comparative analysis of conventional Papanicolaou tests and a fluid-based thin-layer method. Arch Pathol Lab Med. 2003;127:200-4.
- [Google Scholar]
- Performance of a fluid-based, thin-layer papanicolaou smear method in the clinical setting of an independent laboratory and an outpatient screening population in New England. Arch Pathol Lab Med. 1999;123:817-21.
- [Google Scholar]
- Comparison of conventional Papanicolaou smears and a fluid-based, thin-layer system for cervical cancer screening. Obstet Gynecol. 1997;90:278-84.
- [Google Scholar]
- Evaluation and resolution of etiologic factors for unsatisfactory cervical cytology ThinPrep preparations. J Am Soc Cytopathol. 2012;1(1 Suppl):S1-126. [Abstract #225]
- [Google Scholar]
- Not all unsatisfactory ThinPrep cervical Pap tests are unsatisfactory: Reprocessing improves the satisfactory and detection rates of ThinPrep cervical cytology. Diagn Cytopathol. 2010;38:699-701.
- [Google Scholar]
- The unsatisfactory ThinPrep Pap Test: Missed opportunity for disease detection? Am J Clin Pathol. 2002;117:457-63.
- [Google Scholar]
- Reprocessing unsatisfactory ThinPrep Papanicolaou test specimens increases sample adequacy and detection of significant cervicovaginal lesions. Cancer. 2004;102:67-73.
- [Google Scholar]
- Reflex high-risk human papillomavirus DNA testing (Hybrid Capture 2) of bloody ThinPrep specimens with atypical squamous cells of undetermined significance interpretation: Does pretreatment with acetic acid affect test performance? Cancer. 2005;105:452-6.
- [Google Scholar]
- The unsatisfactory ThinPrep® Pap Test™: Analysis of technical aspects, most common causes, and recommendations for improvement. Diagn Cytopathol. 2013;41:588-94.
- [Google Scholar]
- Protocol for the processing of bloody cervical specimens: Glacial acetic acid and the ThinPrep Pap test. Diagn Cytopathol. 2006;34:210-3.
- [Google Scholar]
- A simple method to determine the need for glacial acetic acid treatment of bloody ThinPrep Pap tests before slide processing. Diagn Cytopathol. 2004;31:321-5.
- [Google Scholar]
- Restoring satisfactory status in ThinPrep Pap test specimens with too few squamous cells and containing microscopic red blood cells. Diagn Cytopathol. 2008;36:696-700.
- [Google Scholar]
- The efficacy of reprocessing unsatisfactory cervicovaginal ThinPrep specimens with and without glacial acetic acid: Effect on Hybrid Capture II human papillomavirus testing and clinical follow-up. Am J Clin Pathol. 2002;118:727-32.
- [Google Scholar]
- Clinicopathologic correlation of the unsatisfactory Papanicolaou Smear. Cancer Cytopathol. 1997;81:139-43.
- [Google Scholar]
- Glacial acetic acid treatment and atypical endocervical glandular cells: An analysis of 92 cases. Am J Clin Pathol. 2010;133:799-801.
- [Google Scholar]








